Introduction
One of the important factors driving the recent paradigm shift in the automotive industry is eco-friendliness. In addition, as regulations on greenhouse gas emission are getting more strengthen globally, the demand for improved fuel efficiency in automobiles continues to rise. Concurrently, there is an increasing necessity for sustainable mobility that rely on non-pollutant emitting power sources, the trend in the automotive industry is shifting from combustion engine vehicles to electric vehicles (EVs).1,2 Due to the nature of the electric power source, EVs apply high torque to the tires from the beginning of acceleration, and wear resistance of tire is required due to the high battery load.3,4 Additionally, decreasing rolling resistance of tire is necessary for extending driving distances within the limitation of battery capacity.5 In particular, commercial vehicles have longer driving ranges and carry higher loads than passenger cars, so the demand for improved fuel efficiency due to the transition to electric vehicles is further strengthening.
In 1992, Michelin Tire developed green tire that changed the filler system from carbon black to silica/silane to reduce rolling resistance.6 Research on passenger car radial (PCR) tires has been actively conducted and has recently reached a mature stage.7,8 However, in truck bus radial (TBR) tires, the presence of proteins and lipids in natural rubber (NR), which is used as the base rubber, has the problem of interfering with the silanization and coupling reactions of silica and silane.9 To address this issue in TBR tires, studies are being conducted on epoxidized NR (ENR), which introduces an epoxide groups that have affinity with silica into NR.10,11 This facilitated the formation of chemical bond with silanol groups on the silica surface through ring-opening reaction, effectively resolving reactivity issues.10,11 However, the epoxide group is highly reactive, so a ring opening reaction may occur during mixing, which could potentially result in deterioration of processability and physical properties.12
In this study, silica-filled ENR/BR blend compounds were manufactured according to ENR types and silica contents, and the physical properties and vulcanizates structure of the compound were evaluated. Through this, we aim to understand the physical properties of ENR-25 and ENR-50 according to silica contents and determine the correlation between vulcanizates structure and physical properties.
Experimental Methods
For the experiment, two types of ENR were used as the base rubber: ENR-25 (Epoxyprene 25, Muang Mai Guthrie Public Co., Ltd., Muang Phuket, Thailand) and ENR-50 (Epoxyprene 50, Muang Mai Guthrie Public Co., Ltd., Muang Phuket, Thailand). These were blended with BR (NdBR-60, Mooney viscosity (ML1+4, 100°C): 63MU, Kumho Petrochemical Co., Daejeon, Korea). For the filler, silica (Ultrasil 7000GR, Evonik Industries AG, Essen, Germany, Brunauer–Emmett–Teller (BET) surface area: 160-175 m2/g) was used, and bis-[3-(triethoxysilyl) propyl] tetrasulfide (TESPT, Si-69, Evonik Korea Ltd., Seoul, Korea) used as the silane coupling agent. TDAE oil (Kukdong Oil & Chemicals Co., Yangsan, Korea) was incorporated as a processing aid. Zinc oxide and stearic acid (both from Sigma-Aldrich Corp., Seoul, Korea) were used as crosslinking activators. N-(1,3-dimethylbutyl)-N′-phenyl-phenylenediamine (6PPD, Kumho Petrochemical Co., Daejeon, Korea) and 2,2,4-trimethyl-1,2-dihydroquinoline (TMQ, Sinopec Corp., Beijing, China) were used as antioxidants. Sulfur (Daejung Chemicals & Metals Co., Siheung, Korea) was used as the curing agent. The vulcanization accelerators were cyclohexyl benzothiazole-2-sulfenamide (CBS, 98%, Tokyo Chemical Industry Co. Ltd., Tokyo, Japan) and 1,3-diphenylguanidine (DPG, 98%, Tokyo Chemical Industry Co. Ltd., Tokyo, Japan). N-cyclohexylthio phthalimide (PVI, Shandong Yanggu Huatai Chemical Co., Ltd., Liaocheng, China) was used as a pre-vulcanization inhibitor.
Proton nuclear magnetic resonance spectroscopy (1H NMR; Varian, Unity Plus 300 spectrometer, Garden State Scientific, Morristown, NJ, USA) was used to quantify the epoxide contents in ENR. Samples were dissolved at a concentration of 15 mg/mL in deuterochloroform (CDCl3, Cambridge Isotope Laboratories, Inc., Andover, MA, USA) within a 5 mm NMR tube.
The glass transition temperature (Tg) of ENR was determined using differential scanning calorimetry (DSC-Q10, TA Instruments, DE, USA). The DSC measurements were conducted with a heating rate of 10°C/min, heating the rubber samples from −80°C to 100°C.
To evaluate the degree of filler network formation in the uncured compound, a strain sweep test ranging from 0.28% to 40% was conducted using a rubber process analyzer (RPA 2000, Alpha Technologies, Hudson, Ohio, USA) at a temperature of 60°C. The reduction in storage modulus (G′) with increasing strain indicates the destruction of the filler network. ΔG′ (G′0.28%−G′40%) is commonly known as the Payne effect.13
Mooney viscosity, an evaluation for the processability of compound, was measured using a Mooney rotational viscometer (Vluchem IND Co., Seoul, Korea). It was measured by operating a large rotor (diameter 38.10±0.03 mm, thickness 5.54±0.03 mm) at 100°C for 4 min after preheating for 1 min at 2 rpm according to the conditions specified by the ASTM D1646 condition.
Cure characteristics of the compound were measured using a moving die rheometer (RLR-3-rotorless rheometer, Toyoseiki, Tokyo, Japan) at a temperature of 150°C and a vibration angle of 1 for 30 min. The minimum torque (Tmin), maximum torque (Tmax), and optimum cure time (t90) of the compound can be determined by this experiment. Subsequently, vulcanizates were prepared by vulcanization in a heating press at 150°C using the optimum vulcanization time. For analysis, the vulcanizates were prepared into specimens measuring 10 mm(length) × 10 mm(width) × 2 mm(thickness). These specimens were weighed and then immersed in tetrahydrofuran (THF, 99%, Samchun Chemical Co., Seoul, Korea) and n-hexane (95%, Samchun Chemical Co., Seoul, Korea) at 25°C for 2 days each to remove organic additives inside the specimens. Subsequently, the specimens were dried at 25°C for 1 day. Post-drying, the weight of each specimen was recorded again to calculate the mass fraction of the extracted organic additives. This specimen was then swollen in toluene at 25°C for 1 day and weighed again. Finally, crosslink density was calculated from the measured weight of the specimen using the Flory-Rehner equation (2), as follows:14,15
v: Crosslink density of vulcanizate (mol/g)
Mc: Average molecular weight between crosslink points (g/mol)
Vr: The volume fraction of rubber in the swollen gel at equilibrium
Vs: The molar volume of solvent (cm3/mol)
ρr: The density of the rubber sample (g/cm3)
χ: The polymer–solvent interaction parameter
Additionally, vr0 was determined using the Kraus equation (2). This equation is instrumental in deriving the chemical crosslink density, which represents the crosslink density in the unfilled compound in the Flory-Rehner equation. Further, the degree of filler–rubber interaction, the difference between the total crosslink density (i.e., chemical crosslink density+filler–rubber interaction) and the chemical crosslink density was calculated. This analysis provides a quantitative measure of the filler–rubber interaction.16,17
Vr0: The volume fraction of unfilled rubber in the swollen gel at equilibrium
φ: The volume fraction of filler
The mechanical properties of the vulcanizate, including tensile strength, modulus, and elongation at break, were assessed using dumbbell-shaped specimens with dimensions of 100 mm × 25 mm according to ASTM D412. Measurements were conducted using a universal testing machine (KSU-05M-C, KSU Co., Ansan, Korea) at a speed of 500 mm/min.
Abrasion resistance was evaluated in accordance with ISO 4649 using a Deutsche Industrie Normen (DIN) abrasion tester. A cylindrical sample with a diameter of 16 mm and a thickness of 8 mm, was ground to a load of 5 N against an abrasive sheet on a rotating cylindrical drum at 40 rpm. This process was carried out for 40 s, and the mass loss of the specimen was measured.
The viscoelastic properties of the vulcanizates were measured using two instruments. Initially, a temperature sweep from −80°C to 100°C at a strain of 0.2% and a frequency of 10 Hz was conducted using a dynamic mechanical analyzer (DMA Q800, TA Instrument, DE, USA) to measure tan δ. Subsequently, a strain sweep from 0.5% to 10% at a temperature of 60°C and a frequency of 10 Hz was performed using a dynamic mechanical thermal spectrometer (DMTS, EPLEXOR 500N, GABO, Ahlden, Germany).
Compound formulation was designed to confirm the effect of ENR types and silica contents on the vulcanizates structure and physical properties of the silica filled ENR/BR compounds. Compounds were named based on the ENR types and silica contents and were manufactured using an internal kneader (300cc, MIRAESI Company, Gwangju, Korea). The mixing process and procedure are detailed in Tables 1 and 2, respectively. The fill factor was set to 80%, and the unit was measured in parts per hundred rubber (phr). In the 1st stage, kneading was initiated at 110°C, with a controlled dump temperature between 150°C-155°C. In the 2nd stage began at 50°C, controlling a temperature of 80°C-90°C. After each stage of mixing, the compound was sheeted using a two-roll mill. The optimal cure time was determined using a moving die rheometer (MDR). Then, the optimal cure time was used to manufacture vulcaniztes in a hydraulic press at 150°C.
Results and Discussion
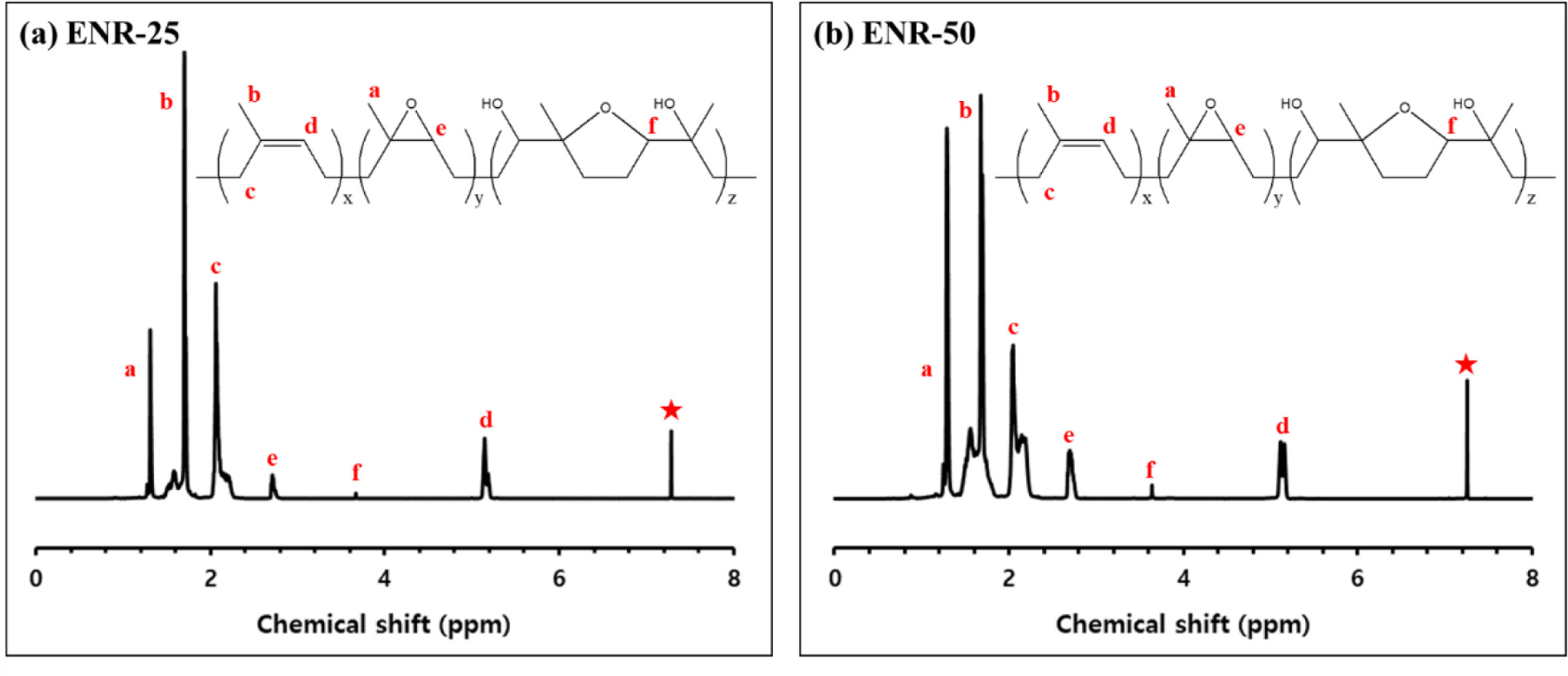
Figure 1 presents the 1H NMR spectra of ENR-25 and ENR-50, while Table 3 lists epoxide contents and Tg of them. In both ENR-25 and ENR-50, characteristic signals were observed: methyl protons (a, b) at 1.37 ppm and 1.84 ppm, methylene protons (c) at 2.3 ppm, methine protons (d) at 5.1 ppm, epoxide protons (e) at 2.7 ppm, and solvent peak of CDCl3 (✶) at 7.26 ppm. Notably, at 3.6 ppm, proton signals (f) indicative of the cyclic ether structure were generated by the ring-opening of adjacent epoxide groups.18 The epoxide contents were quantified using the peak areas at 2.7 ppm and 5.1 ppm (A2.7, A5.1).19
| Rubber | 1H NMR results | DSC results |
|---|---|---|
| Epoxide contents (mol %) | Tg (°C) | |
| ENR-25 | 25 | −42 |
| ENR-50 | 46 | −25 |
As a result, ENR-25 contained 25 mol % epoxide groups, while ENR-50 contained 46 mol %. Tg was influenced by the limited chain mobility due to increased intermolecular interactions as polarity of chain increased.20,21 Consequently, ENR-50, with its higher epoxide contents, exhibited a Tg of −25°C, compared to −42°C for ENR-25.
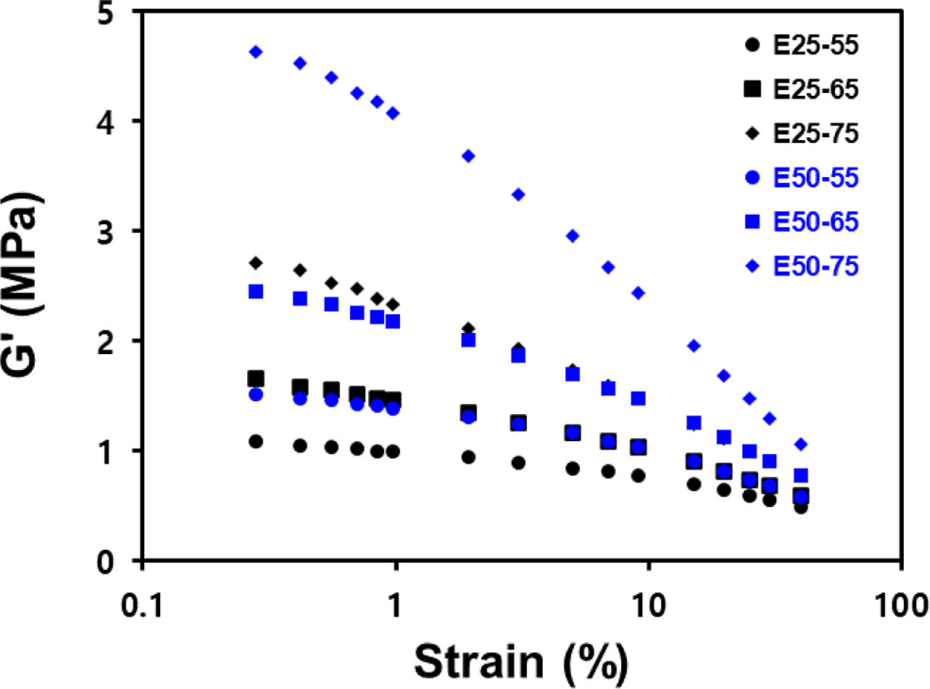
Figure 2 and Table 4 display the Payne effect measurements of the compounds. The results indicated that the ENR-50/BR blend compounds exhibited higher ΔG′ values than the ENR-25/BR blend compounds at equivalent silica contents. This difference is attributed to the formation of strong hydrogen bonds between ENR molecules in ENR-50 due to its high epoxide contents. These bonds tend to break as strain amplitude increases.10 Furthermore, silica-filled ENR/BR blend compounds showed increased filler–filler interaction with increasing silica contents, leading to high ΔG′.
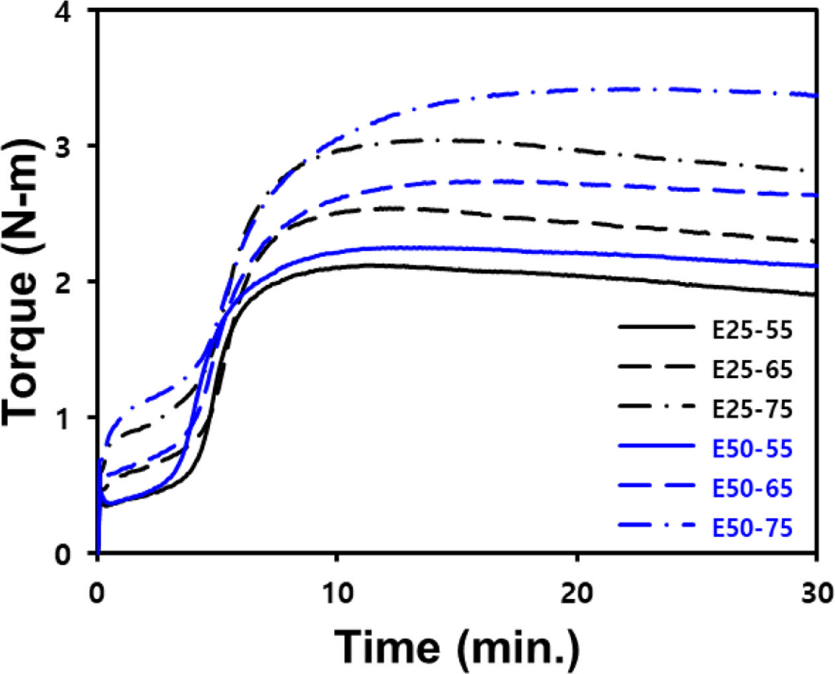
The cure characteristics obtained using MDR and Mooney viscosity, a measure of compound processability, are shown in Figure 3 and Table 4. ENR-50/BR blend compounds exhibited higher Mooney viscosity than the ENR-25/BR blend compounds at the same silica contents. This trend is attributed to the greater occurrence of ring-opening reaction of the epoxide group during mixing. Additionally, there was a tendency for Mooney viscosity to increase with the silica contents.
In the analysis of MDR curves, the ΔT (Tmax−Tmin) is closely correlated with the crosslink density of the compound.22 ENR-50 forms a stronger filler–rubber interaction with silica than ENR-25, so it exhibits a higher ΔT. Silica can adsorb vulcanization accelerators because it is a hydrophilic filler.23 Additionally, ENR has polar characteristics that contribute to the adsorption of vulcanization accelerators.11 Consequently, the longest t90 was observed in E50-75 compound, which contain the highest silica contents and use ENR-50.
Table 5 presents the results from the analysis of the vulcanizates structure. Total crosslink density of the compound was analyzed by dividing it into chemical crosslink density and filler–rubber interaction through the filler variation. Chemical crosslink density reflects the network formed between rubber chains through the action of a curing agent or the functional groups within the rubber. In contrast, filler– rubber interaction represents crosslink density formed by the physical and chemical bonds between the filler and rubber. ENR-50 has superior reactivity to silanol groups compared to ENR-25, and at the same time, ring opening reaction between base rubbers can also occur actively. Furthermore, it was observed that increasing the silica contents in ENR-50/BR blend compounds led to a further improvement in filler–rubber interaction compared to the ENR-25/BR blend compounds.
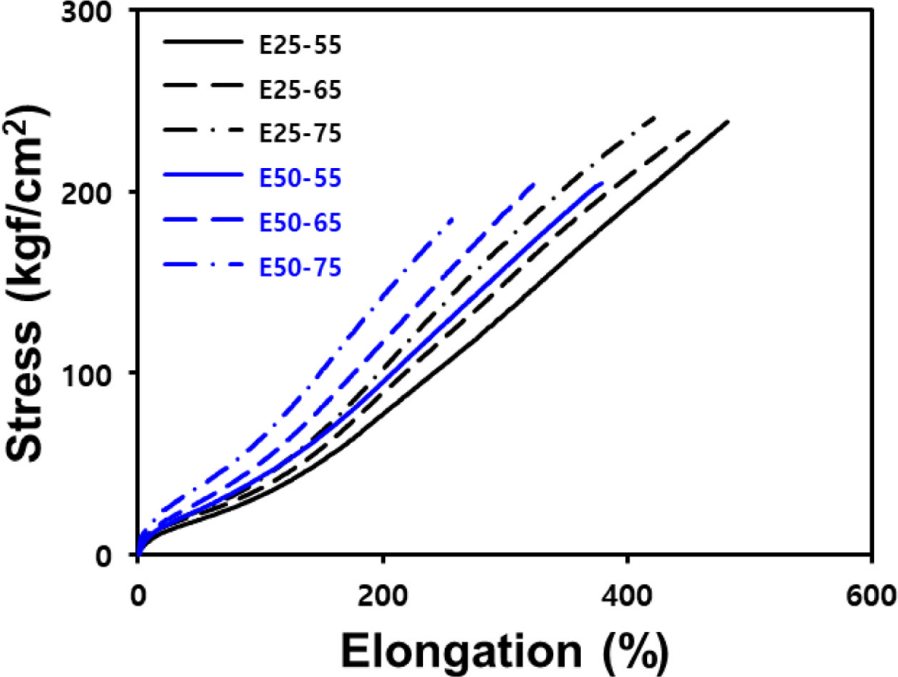
Mechanical properties are a measure of rubber stiffness and exhibit a strong correlation with crosslink density.24 ENR-50/BR blend compounds, which has higher crosslink density, showed higher tensile modulus compared to ENR-25/BR blend compounds. Furthermore, an increase in silica contents not only enhanced the modulus due to improved reinforcement properties but also led to a reduction in elongation. Consequently, it was observed that toughness of silica-filled ENR/BR blend compounds decreased with increasing silica contents.
In the DIN abrasion test, abrasion loss is closely correlated with filler–rubber interaction and toughness.25,26 The results showed that while an increase in filler contents strengthened filler–rubber interaction, it concurrently reduced toughness. Consequently, ENR-50/BR blend compounds generally exhibited better abrasion resistance due to their superior filler–rubber interaction compared to ENR-25/BR blends. Notably, the E50-65 compounds exhibited the best abrasion resistance.
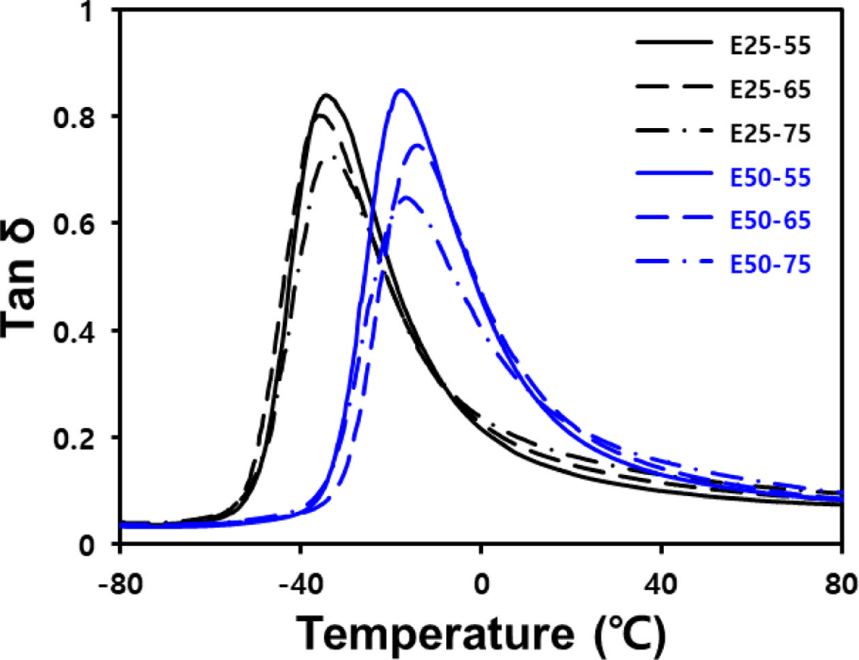
When a tire rotates with the vehicle load applied, all parts of the tire undergo repeated deformation and recovery, causing energy loss. This phenomenon is called hysteresis loss. In the low-temperature region, where the motion of filler is restricted, micro-brownian motion of the polymer within the network is the main source of energy dissipation.27 Therefore, in the vicinity of Tg, hysteresis loss of the compound increases with increasing volume fraction of the polymer. Tan δ at 60°C serves as an indicator of hysteresis loss due to the destruction and reformation of the filler network. A higher tan δ suggests an undesirable rolling resistance for the compound.28 E” at 0°C is indicative of wet grip performance, with higher values correlating with better performance.29
Figure 5 and Table 7 illustrate the variation in tan δ with temperature changes, as measured using DMA at a 0.2% strain. In the silica-filled ENR/BR blend compounds, as the silica contents increase, the polymer volume fraction decreases, resulting in lower tan δ at Tg. Additionally, hysteresis loss increases by forming more silica network. Furthermore, it has been observed that a higher Tg of the polymer used can enhance wet grip performance, but it is disadvantageous to fuel efficiency.30 Therefore, E50-75 compound exhibited the highest E” at 0°C and tan δ at 60°C.
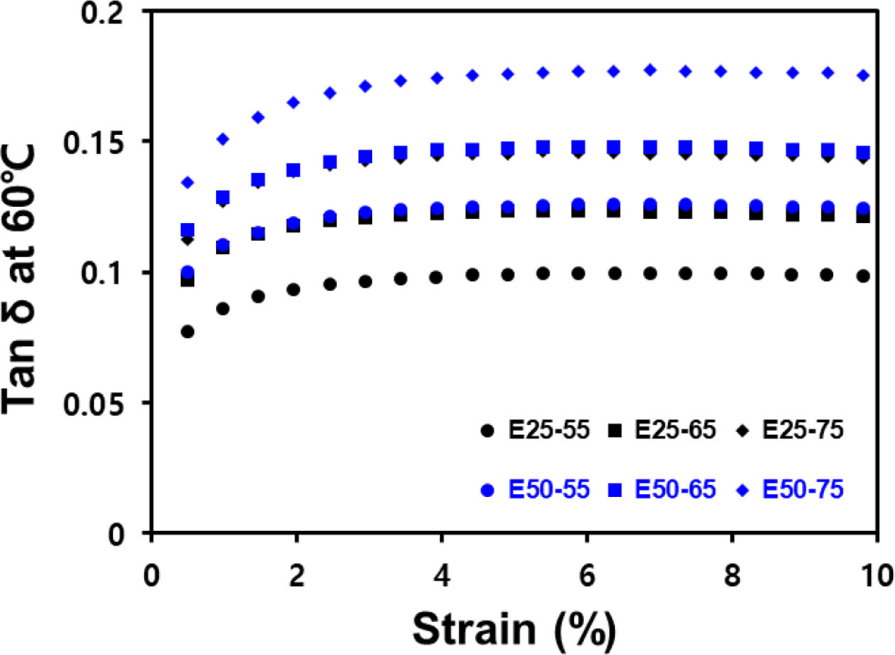
Figure 6 and Table 7 present the tan δ value according to strain changes at 60°C, using DMTS. In condition of 5% or higher strain, filler network is mostly destructed, so the effect of hysteresis due to the destruction and reformation of filler network appears significantly.31,32 Consequently, an increase in silica contents was found to correlate with increased hysteresis. Notably, the E25-55 compounds demonstrated the lowest tan δ at 60°C.
Conclusion
In this study, silica-filled ENR/BR blend compounds were manufactured and evaluated to determine the effect of ENR types and silica contents on the physical properties and vulcanizates structure. It was confirmed that when the contents of silica that can react with ENR during mixing increased, processability of the compound decreased significantly and the filler–filler interaction was formed strongly. ENR-50 which has higher epoxide contents, exhibited stronger filler– rubber interaction compared to ENR-25, leading to superior abrasion resistance. Additionally, Due to the high Tg of ENR-50, wet grip performance was improved, but rolling resistance was increased. Furthermore, increasing the amount of silica had little effect on abrasion resistance due to increased filler–rubber interaction and decreased toughness. But, it did contribute to improved wet grip performance and increased rolling resistance. As a result, it was confirmed that TBR tire tread compound can be designed by applying ENR-50 to improve wear resistance and wet grip performance, and by applying ENR-25 and reducing silica contents to improve fuel efficiency. This study provides guidelines for introducing ENR to apply silica/silane system in TBR tire tread compound and direction for optimizing compounds to achieve desired performance levels with varying silica contents.