Introduction
Tire wear particles (TWPs) are generated by friction between the tire tread and the road surface during vehicle movement. The particle size of TWPs ranges from several hundred micrometers to approximately 1 μm.1 In the road environment, TWPs are typically encapsulated by mineral particles forming tire-road wear particle (TRWP) composites. The global annual generation of TWPs is estimated to exceed 6 million tons.2 Owing to the significant contribution to particulate emissions, the Euro 7 regulation is the first to introduce standards not only for exhaust emissions but also for non-exhaust particles, with an anticipated reduction in particulate emissions by approximately 10-30%. Moreover, the growing adoption of electric vehicles, generally heavier than conventional vehicles, is expected to further exacerbate TWP generation, highlighting the need for effective TWP mitigation strategies. In this context, attention has increasingly focused on the environmental impacts of nonexhaust emissions such as TWPs.3
Leachates from TWPs have been reported to disrupt thyroid hormone regulation and induce fibrotic damage in lung tissue.4,5 The antioxidant 6PPD, commonly used in tire formulations, undergoes oxidation by ozone and ultraviolet radiation to form 6PPD quinone, a compound identified as an ecotoxic in aquatic environments.6,7 Following their generation, TRWPs are either dispersed into aquatic environments or become airborne, subsequently embedding on surrounding soil and the atmosphere.8-10 The average density of TRWPs has been reported to increase slightly with decreasing particle size, suggesting that finer particles tend to be more compact or more heavily embedded with mineral components.11 Accordingly, the size and density of TWPs influence their gravitational settling velocities.12
Road dust is repeatedly resuspended by passing traffic, complicating the isolation and study of primary wear particles. By contrast, TRWPs deposited on snowbanks formed by mechanical snow clearing remain immobilized, preventing further resuspension. Consequently, road dust collected from snow offers a unique opportunity to investigate the characteristics of single-step scattering of TRWP emissions immediately after their generation. On urban roads, the typical speed limit is 50 km h−1, and vehicles frequently encounter low-speed conditions owing to stopping at traffic signals and gradual acceleration.13 Additionally, wet conditions in the winter reduce friction between the tire tread and asphalt surface.14 In this study, road dust deposited on snow by traffic scattering was collected at a pedestrian crossing, approximately 5 m from the crossing and at lateral distances of 0, 0.7, and 1.0 m from the roadside, where vehicles typically travel at low speeds.
Quantification of the TWP content in environmental samples is generally based on the analysis of chemical markers specific to TWPs, including natural rubber (NR), butadiene rubber (BR), styrene-butadiene rubber (SBR), zinc, sulfur, and bound sulfur.15-18 Qualitative and quantitative analyses of TWPs in road dust samples have been performed using pyrolysis-gas chromatography/mass spectrometry (Py-GC/MS), employing key pyrolysis products of NR, BR, and SBR as chemical markers. The use of rubber markers to determine the TWP content in road dust helps minimize analytical errors compared to other analytical techniques, as rubber components constitute the largest proportion (approximately 50%) of tire tread compounds.19-22 This study aimed to investigate the distribution and characteristics of TRWPs in road dust collected under wet winter road conditions and low-speed driving environments, focusing on the variations according to the distance from the roadside.
Experimental
Road dust samples were collected from a snow-covered area on the side beyond the crosswalk (37°32′56.8″N, 127°4′31.3″E), at distances of 0, 0.7, and 1.0 m from the roadside edge (Figure 1). The sampling was conducted in the afternoon of February 11, 2025, on a clear day following snowfall events of 3.3 cm and 0.7 cm that occurred on February 6 and 7, respectively. Vehicles in the slow lanes operated under mild driving conditions at speeds below 50 km h−1. Sampling was conducted on the snow because it contained only road dust, including TRWPs dispersed from the road surface. To examine the variation in particle dispersion as a function of distance from the road, samples were collected at the three specified points.

Following the measurement of the sampled snow weight, the samples were transferred to petri dishes and dried in a convection oven at 70°C for 24 h. The remaining solid component was then scraped off using a spatula and weighed. The solid sample (road dust) was size-separated using an Octagon 200 sieve shaker (Endecotts Co., UK). Standard sieves with apertures of 1000, 500, 212, 106, 63, 38, and 20 μm (Endecotts Co., UK) were used, and size separation was performed in interval mode for 20 min.
Rubber composition of TWPs in the road dust samples was determined by analyzing main pyrolysis products formed from NR, BR, and SBR using Py-GC/MS. Py-GC/ MS analysis was carried out using an Agilent 6890 gas chromatograph equipped with a 5973 mass spectrometer (Agilent Technology Inc., USA) equipped with a JCI-55 Curie point pyrolyzer (Japan Analytical Industry Co., Japan). A pyrofoil of 590°C Curie temperature was used. Pyrolysis was performed for 10 s using the pyrofoil under helium (He) atmosphere. A DB-5MS capillary column (30 m× 0.32 mm, 25 μm film thickness) (Agilent Technology Inc., USA) was used. The injector temperature was 250°C. The GC oven temperature program was as follows: 30°C (held for 3 min), increased to 50°C at 10°C min−1 (held for 3 min), then to 180°C at 10°C min−1 (held for 1 min), and finally to 250°C at 10°C min−1 (held for 3 min). The interface temperature of GC to MS was 250°C. The electron ionization (70 eV) was used to ionize the pyrolysis products. The MS source temperature was 230°C.
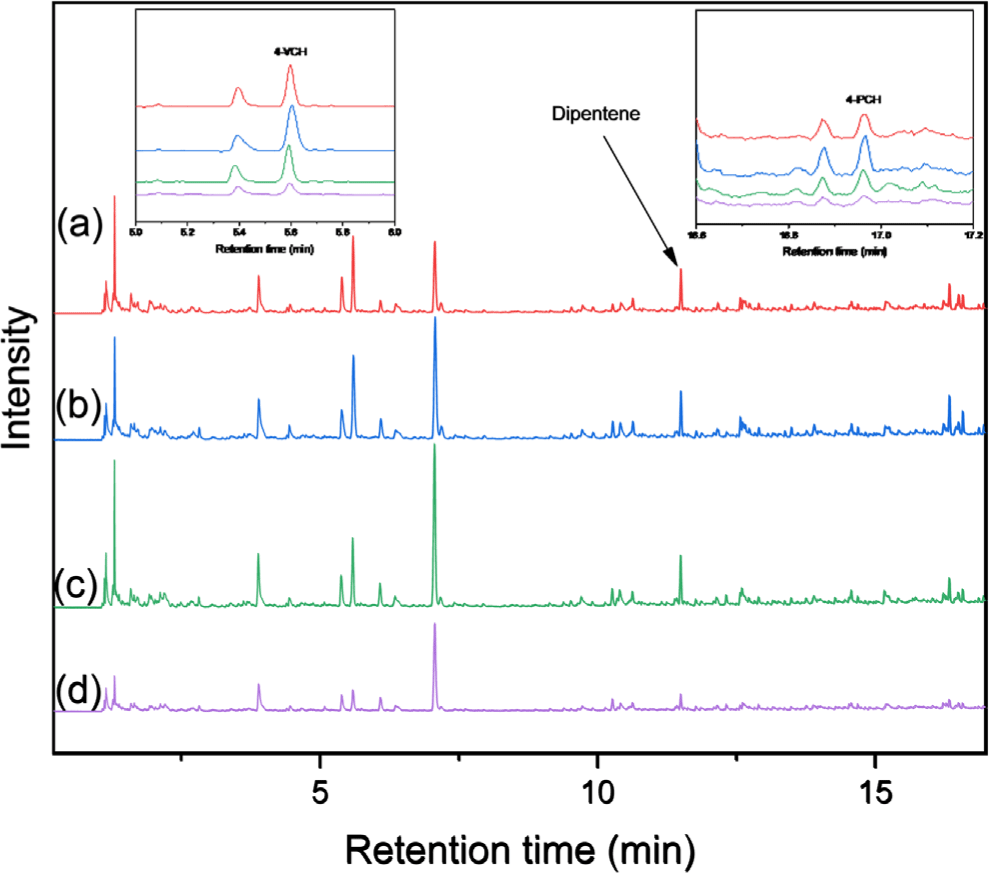
Standard Malaysian rubber of SMR CV60, BR01 (Kumho Petrochemical Co., Republic of Korea), and SBR1502 (Kumho Petrochemical Co., Republic of Korea) were used as reference NR, BR, and SBR, respectively, to build calibration curves. For building the calibration curves, the raw rubber was completely dissolved in toluene, and the solutions of 0.1, 0.25, 0.5, 1.0 and 1.5 mg rubber mL-1 were prepared. The standard sample preparation process for the calibration curve was as follows: (1) drop 20 μL of the rubber solution on the slide glass, (2) evaporate the solvent in a convection oven at 70°C for 30 min, and (3) use a scalpel to shape the dried rubber into a ball and place it in the center of the pyrofoil. The reference samples of 20 μL of each solution corresponding to the rubber weights of 2, 5, 10, 20 and 30 μg, respectively, were analyzed. The calibration curves were built using dipentene, 4-vinylcyclohexene (4-VCH), and 4-phenylcyclohexene (4-PCH) for quantification of NR, BR, and SBR respectively. The calibration curves had good linearity (Figure 2).
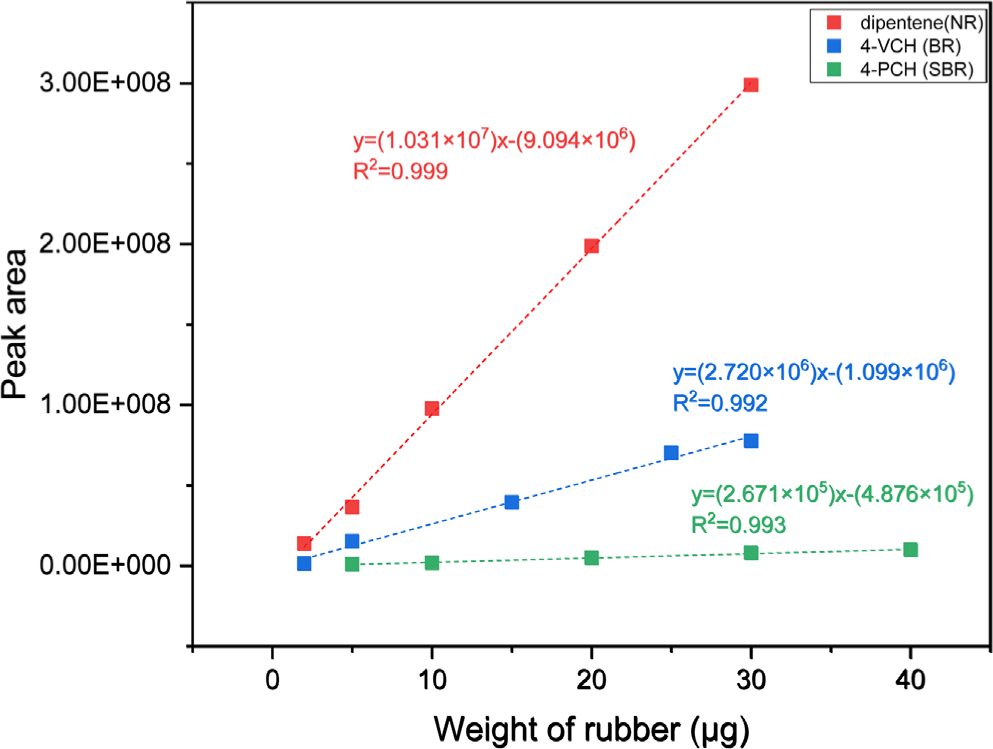
Presence of SBR in the road dust samples was determined by detection of 4-PCH. Concentrations of NR, BR, and SBR were determined using the calibration curves and the peak areas. The peak area ratio of 4-PCH to 4-VCH in SBR1502 was approximately 0.254. Therefore, when both 4-VCH and 4-PCH were identified in the road dust samples, the peak area of 4-VCH from BR was calculated by subtracting the peak area of 4-PCH divided by 0.254 from the total peak area of all 4-VCH peak area.
Results and Discussion
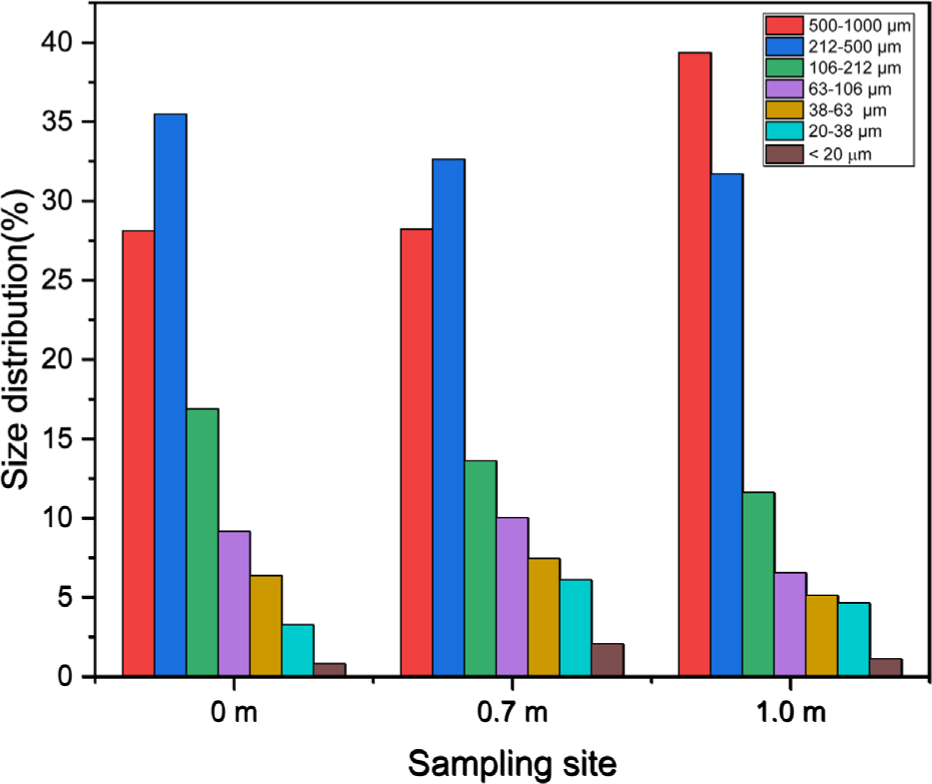
The snow collected at the sampling sites located 0, 0.7, and 1.0 m from the roadside were 11.49, 12.74, and 11.32 g, respectively. The corresponding solid contents were 299, 441, and 282 mg, respectively. The sampling locations were sufficiently distant from surrounding natural sources, and no weather events, such as yellow dust, occurred during the collection period. Therefore, the solid components were attributed to road dust transferred from the road surface by the traffic. Figure 3 shows the particle size distributions of the solid components, excluding particles larger than 1000 μm. The dominant size range was 212-1000 μm, accounting for 63, 60, and 71% of the total mass at the 0, 0.7, and 1.0 m sampling sites, respectively. For the road dust on dry road, the dominant particle ranges tended to shift to smaller size.23 However, the particle size distribution of road dust is affected by various conditions like the road conditions, traffic volume, and vehicle speed. The proportion of road dust in the 500-1000 μm size range tended to increase with distance from the roadside, whereas that of particles in the 106-500 μm range decreased with increasing distance. The particles smaller than 106 μm were most abundant at the 0.7 m location.
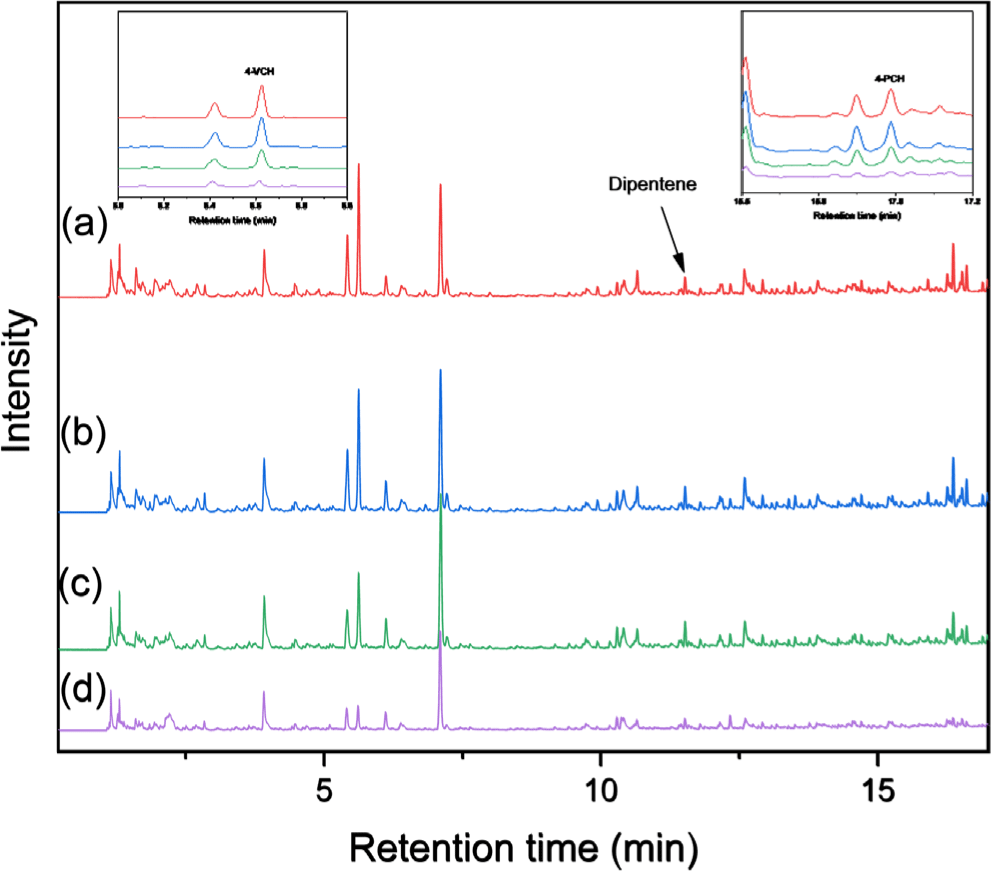
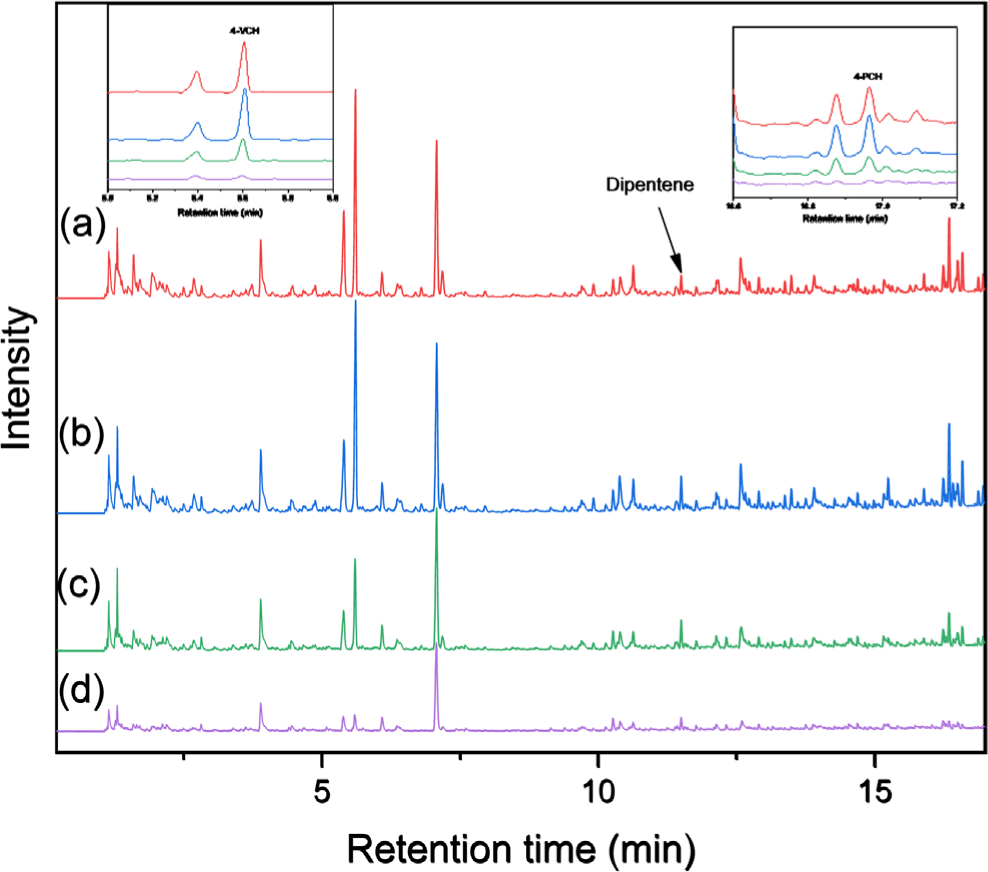
The rubber composition and TWP concentration in the road dust fractions smaller than 212 μm were analyzed using Py-GC/MS. The Py-GC/MS chromatograms of the samples collected at 0, 0.7, and 1 m are shown in Figures 4, 5, and 6, respectively. Typical pyrolysis products of NR, BR, and SBR were identified in the chromatograms: dipentene was identified as a key pyrolysis product of NR; 4-VCH was a key pyrolysis product derived from BR and the butadiene sequence of SBR; and 4-PCH was a unique pyrolysis product generated from the styrene-butadiene heterosequence of SBR.20-22 Additionally, 2-phenylpropene (2-PP) and 3-phenylcyclopentene (3-PCP) were obtained as the pyrolysis products from the styrene-butadiene heterosequence of SBR. These results confirmed that three rubber components of NR, BR, and SBR used for tire tread compounds were present in the samples.
The rubber composition varied depending on particle size and sampling location (Figure 7). The rubber composition of the 0 m sample showed a distinct trend: the NR content decreased significantly from 41% to 7% as the particle size decreased from 106-212 μm to 20-38 μm. By contrast, the SBR ratio increased from 25% to 93%, and the BR proportion decreased from 34% to 0%. In the 0.7 m sample, SBR was the dominant rubber type; however, no clear trend with particle size was observed. The rubber composition of the 1.0 m sample was similar to that of the 0 m sample but exhibited smoother trends: the NR proportion decreased from 55% to 35% as the particle size decreased from 106-212 μm to 20-38 μm, the SBR ratio increased from 24% to 55%, and the BR proportion decreased from 21% to 10%.
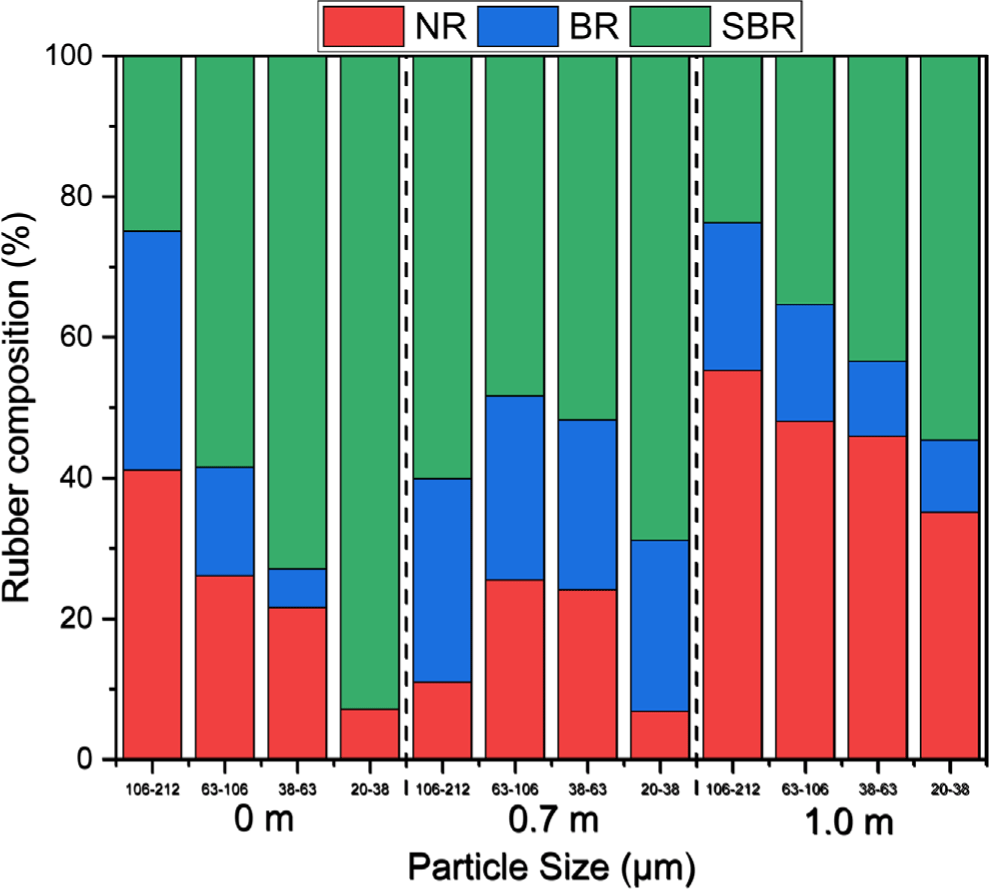
The average rubber compositions were NR/BR/SBR = 24.0/13.7/62.3, 16.8/25.9/57.3, and 46.0/14.6/39.4 for the sampling locations at 0, 0.7, and 1.0 m, respectively. SBR was the most abundant rubber component irrespective of the sampling location, and NR was the second most abundant, except at 0.7 m. The average rubber composition across the three samples was NR/BR/SBR = 28.9/18.1/53.0. These results indicate that SBR was the dominant rubber component in TWPs. NR is the primary rubber in tire tread compounds of buses and trucks, whereas SBR is primarily used in passenger car tire treads. Thus, most of the TWPs in the collected road samples can be concluded to originate from the abrasion of passenger car tire treads.
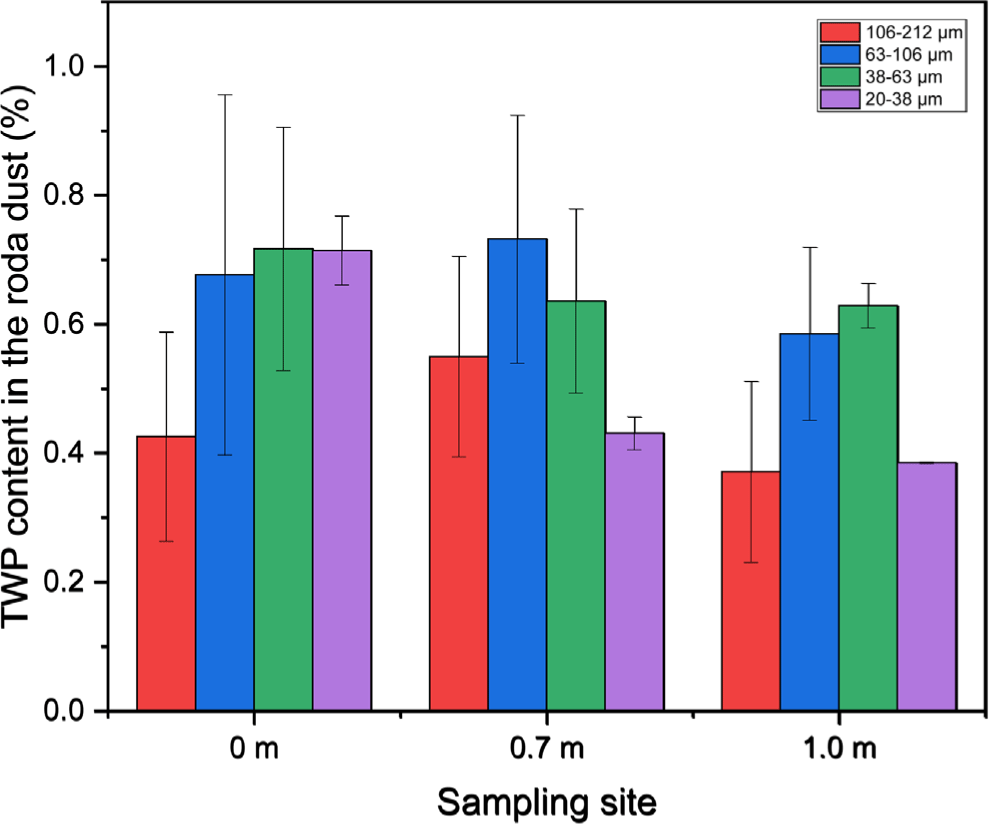
To quantify the TWP concentration in the road dust samples, the rubber weight was converted to the tire compound weight using an average tire tread formulation (rubber: 100 phr; filler: 80 phr; and additives: 20 phr). The conversion factor was 200/100 = 2.00. The TWP concentrations are shown in Figure 8. The TWP contents did not exceed 1%, irrespective of the sampling location or particle size. The trends in TWP content concerning particle size were as follows: 38-63 μm to 20-38 μm > 63-106 μm > 106-212 μm for the 0 m sample; 63-106 μm > 38-63 μm > 106 - 212 μm > 20 - 38 μm for the 0.7 m sample; and 38-63 μm > 63-106 μm > 20-38 μm to 106-212 μm for the 1.0 m sample. The 38-106 μm particle size range exhibited the highest TWP content at all sampling locations. The TWP content decreased for the 20-38 μm fraction as the distance of sampling location from the roadside increased.
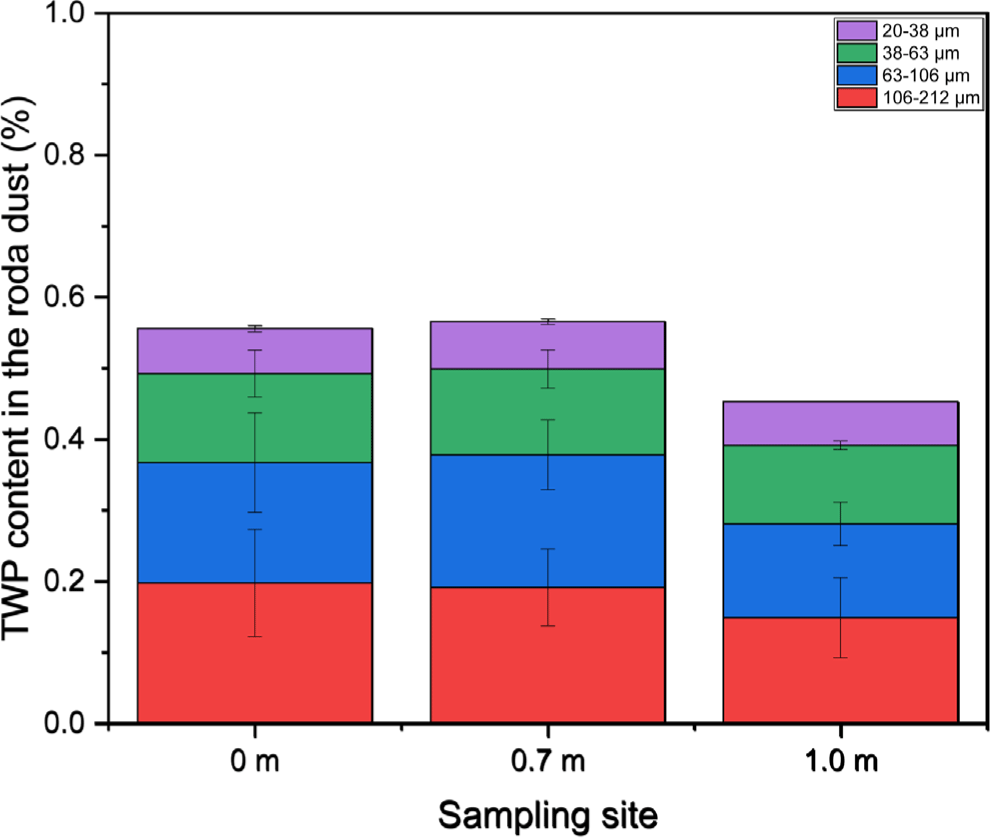
The TWP concentration in the 20-212 μm range was calculated considering the particle size distribution (Figure 9). The TWP contents were 0.56, 0.57, and 0.45% at 0, 0.7, and 1.0 m, respectively. The TWP contents at 0 and 0.7 m were nearly identical and larger than those at 1.0 m. The TWP contents in the road dust transferred from wet roads were lower than those obtained from dry roads. A previous study reported approximately 1% TWP content in road dust collected from a dry road. This reduction may be attributed to road dust being washed away from the road surface by snow melt. Because TRWPs have a lower density than mineral particles, they are more likely to be washed away than mineral particles.
Conclusions
This study systematically analyzed the particle size distribution and rubber composition of TWPs in road dust samples transferred by single-step scattering at different distances from the roadside, using road dust deposited on snow. The road dust particles in the 500-1000 μm size range tended to increase with distance from the roadside, whereas those in the 106-500 μm range decreased with increasing distance. Three major rubber components, NR, BR, and SBR, used in tire tread compounds, were identified in the samples. The NR and BR fractions tended to decrease with decreasing particle size, whereas the SBR proportion increased. SBR was the most abundant rubber component, followed by NR, indicating that the TWPs from passenger cars were more prevalent than those from buses and trucks. The TWP contents in the 20-212 μm road dust samples were 0.56, 0.57, and 0.45% at 0, 0.7, and 1.0 m, respectively, which were lower than those obtained from the dry roads. This is owing to the washing away of road dust by snow melt.






