Introduction
Lithium-ion batteries (LIBs) have been widely utilized in various fields such as electric vehicles, energy storage systems, and portable electronics, establishing themselves as an essential technology in the modern energy industry.1,2 With their high energy density, long cycle life, and excellent stability, LIBs are regarded as the standard for next-generation energy storage devices.3
A lithium-ion battery (LIB) is fundamentally composed of a cathode, an anode, an electrolyte, a separator, and a binder.4 Each component interacts closely with the others to determine the electrochemical performance and lifespan of the cell. The cathode determines the energy density through its high operating voltage and structural stability, while the anode governs the battery capacity by accommodating lithium ions during discharge.5 The electrolyte serves as a pathway for lithium-ion transport, determining the ionic conductivity between electrodes.6 The separator prevents direct contact between the cathode and anode, allowing only ions to pass through selectively and thus avoiding electrical short circuits.7 The binder binds active materials and conductive additives to the current collector.8,9
The binder plays a vital role in ensuring both the electrochemical performance and mechanical stability of the battery by maintaining the cohesion of active particles within the electrode.10 It preserves the structural integrity and mechanical robustness of the electrode during the volume fluctuations that occur throughout charge/discharge cycling.11 In addition, the binder influences electrochemical efficiency by enhancing interfacial stability with the electrolyte, facilitating lithium-ion transport, and promoting uniform SEI formation.12
Currently, the most widely used binders in commercial lithium-ion batteries (LIBs) include poly(vinylidene fluoride) (PVDF), polytetrafluoroethylene (PTFE), carboxymethyl cellulose (CMC), styrene–butadiene rubber (SBR), and poly(acrylic acid) (PAA).13,14 These binders have been optimized for large-scale manufacturing, offering high reliability and reproducibility. PVDF provides high adhesion to metal current collectors and excellent chemical and mechanical stability but has limitations such as electrical insulation properties and the need to use NMP, a toxic solvent.15 In contrast, water-based systems such as CMC/ SBR and PAA are environmentally friendly but exhibit limited mechanical strength and thermal stability, leading to structural deformation in high-energy or high-temperature conditions.12 Other synthetic and natural polymer binders, including poly(vinyl alcohol) (PVA),16 polyacrylamide (PAM),17 sodium alginate (SA),18 and guar gum (GG),19 have also been explored. However, challenges remain in achieving sufficient cycling stability, adhesion strength, and interfacial compatibility.20 Therefore, across both cathodes and anodes, binders must simultaneously enhance mechanical durability and interfacial stability.21
Polyimide (PI) is a polymer with excellent thermal stability, mechanical strength, and chemical resistance, which has been applied in various fields such as aerospace, electronic devices, and lithium-ion battery electrodes.22–24 The resonance-stabilized imide ring and aromatic backbone allow PI to retain structural stability even above 400°C, making it suitable for high-temperature electrode fabrication.20 Moreover, the molecular structure of polyimide can be finely synthesized through the choice of monomers and functional groups, allowing customized designs to meet the specific demands of each electrode system.25
This review focuses on the design strategies of polyimide-based binders and provides a comprehensive overview of their applications across various electrode systems, including cathodes (LFP and NCM) and anodes (graphite and silicon). It discusses how the molecular and structural design principles of polyimides can be optimized to meet the distinct mechanical and electrochemical requirements of different electrode materials.
Design Strategies of Polyimide Binders
Polyimide (PI) is generally synthesized through the condensation reaction between a dianhydride and a diamine to form a poly(amic acid) (PAA) precursor, which is then converted into PI via thermal or chemical imidization (Figure 1).26-28 The imidization reaction involves dehydration condensation, where the –COOH and –NH– groups within the amic acid form a cyclic imide structure.29 The reaction rate and the resulting polymer structure depend on factors such as temperature, reaction time, and the electronic characteristics of the monomers.30,31 Organic solvents such as N-Methyl-2-pyrrolidone (NMP) and dimethylformamide (DMF) are used to ensure high solubility and homogeneous polymer chain formation.32 However, these processes using organic solvents impose significant environmental burdens due to their toxicity and wastewater treatment issues, thereby limiting scalability for large-scale and eco-friendly production.33
To address these concerns, water-based synthesis methods have recently emerged as an environmentally sustainable alternative. This approach involves introducing hydrophilic monomers such as 3,5-diaminobenzoic acid (DABA) or converting the poly(amic acid) precursor into a salt form to make it dispersible in water.34 Such aqueous processes minimize the use of toxic organic solvents while preserving the intrinsic thermal stability and mechanical strength of polyimide, making them a key technology for developing eco-friendly polymer binders.35
Polyimide can be precisely tailored through monomer combination and functional group substitution strategies, allowing balanced control of polymer rigidity, flexibility, and functionality. For example, carboxyl groups (–COOH) improve adhesion to electrode particles by forming hydrogen bonds and ester linkages with the surfaces of silicon or metal oxides.36 Ether groups (–O–) enhance the flexibility of the polymer backbone by increasing its rotational freedom, which facilitates lithium-ion transport within the electrode.37 Sulfonic acid groups (–SO3H) create ionic conduction pathways in the polymer, thereby improving ionic conductivity.38 Meanwhile, trifluoromethyl groups (–CF3) enhance dielectric stability and reduce moisture absorption, contributing to the long-term stability of the electrode.39
In this way, the structural design of polyimides not only enhances adhesion but also stabilizes the electrode–electrolyte interface, strengthens the conductive network, and alleviates internal stress during charge–discharge processes.40 Such molecular-level engineering provides an effective design strategy for developing high-performance multifunctional binders.4
Polyimide Binder for Cathodes
In lithium-ion batteries (LIBs), the most widely used representative cathode materials are lithium iron phosphate (LiFePO4, LFP) and nickel-cobalt-manganese oxide (LiNixCoyMnzO2, NCM).41 LFP has a theoretical capacity of 170 mAh g−1, but its limited Li+ diffusion and low electronic conductivity lead to polarization and low rate capability.42 In contrast, NCM cathodes operate at higher voltages of 4.2-4.4 V and deliver discharge capacities above 200 mAh g−1, offering significantly higher energy density than LFP.43 However, their layered oxide structure requires careful management under high-voltage operations to maintain interfacial and structural stability.44
In this section, the design strategies of polyimide (PI) binders and their effects on enhancing the electrochemical performance of LFP and NCM cathodes are discussed.
To enhance the performance of LFP electrodes, it is essential to strengthen the electrical and mechanical connectivity between particles and ensure interfacial stability among electrode components.45 Accordingly, studies on polyimide (PI) binders in LFP electrodes have focused on achieving strong interparticle adhesion and stable ion/electron transport pathways, thereby improving both structural and electrochemical stability.46
According to a recent study, Zhang et al. (2020) synthesized a redox-active polyimide (PI) binder via hydrothermal polymerization using 3,3′,4,4′-benzophenonetetracarboxylic dianhydride (BTDA) and 1-(4-aminophenyl)-2,3-dihydro-1,3,3-trimethyl-1H-indene-5-amine (DAPI) and applied it to LFP electrodes (Figure 2a).47 The resulting PI binder enhanced particle dispersion and adhesion strength, while its intrinsic ionic conductivity increased the lithium-ion diffusion coefficient (Dli+) to 8.0 × 10−14 cm2 s−1 (Figure 2b). Moreover, the carbonyl groups (–C=O) within the polyimide chains participated in reversible enolization reactions, contributing to additional charge storage. As a result, the electrode exhibited 98 % capacity retention after 200 cycles (Figure 2c), outperforming PVDF-based electrodes in electro-chemical performance.
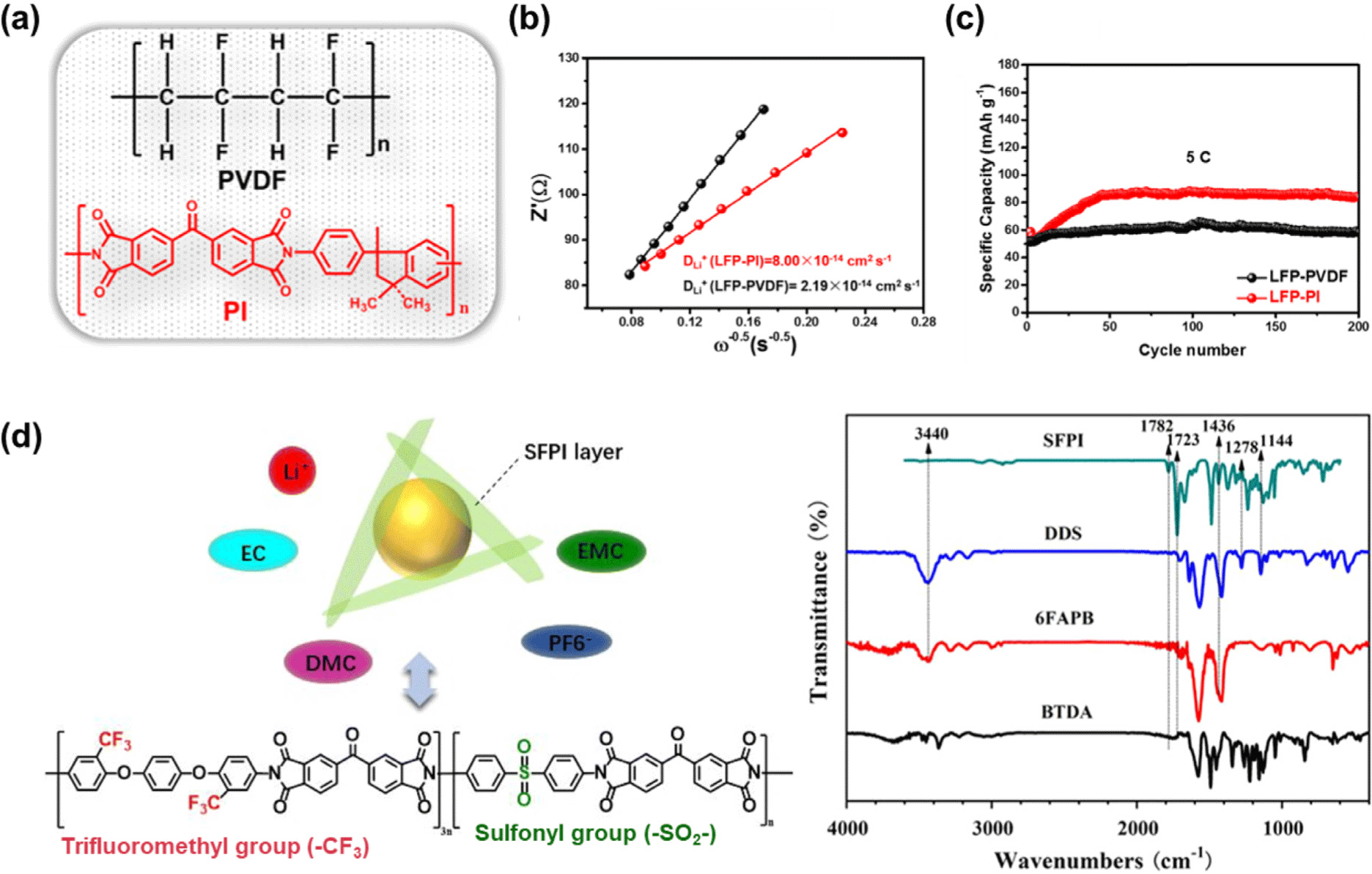
Subsequently, Liu et al. (2021) designed a fluorinated polyimide (SFPI) binder incorporating fluorine (–CF3) and sulfone (–SO2–) functional groups and applied it to LFP electrodes (Figure 2d).48 The SFPI binder exhibited significantly improved adhesion to the active material surface due to strong polar interactions between the functional groups, leading to reduced impedance and polarization and thus more efficient electron and ion transport. Consequently, the SFPI-based LFP electrode delivered a discharge capacity of 134.1 mAh g−1 and maintained 94.1% of its capacity after 100 cycles. Owing to its superior thermal stability and electrochemical durability, it also operated stably under overcharge conditions. In particular, the incorporation of –CF3 and –SO2– groups enhanced both electrolyte affinity and interfacial stability, effectively enabling the realization of highly safe and durable LFP electrodes.
Polyimide (PI) binders incorporating functional groups enhance the mechanical integrity, ionic transport, and interfacial stability of LFP electrodes, leading to improved capacity retention and thermal durability.
At high operating voltages, NCM electrodes suffer from metal dissolution and structural degradation due to strong oxidative reactivity, leading to reduced long-term stability.44,49,50 To mitigate these issues, strengthening interparticle bonding and stabilizing the electrode–electrolyte interface is crucial, and many researchers have pursued this using PI binders.51
Qi et al. designed aromatic polyimide binders, PI(OBO) and PI(BBP), incorporating both soft (ether-linked) and hard (biphenyl-based) segments and applied them to NCM811 cathodes (Figure 3a). The ether-based soft segments enhanced Li+ affinity, thereby improving ion transport and interfacial adhesion, while the rigid aromatic segments provided excellent thermal resistance, effectively reinforcing the thermal stability of NCM811 cathodes. As a result, they demonstrated excellent rate capabilities at 5 C in the voltage range of 2.5-4.3 V, delivering 138.0 mAh g−1 for PI(OBO) and 127.7 mAh g−1 for PI(BBP). Moreover, after 100 cycles at 4.5 V, capacity retention reached 39% for PI(OBO) and 62% for PI(BBP), showing substantial improvement over PVDF. The formation of a uniform protective layer on particle surfaces contributed to CEI stabilization and suppression of structural degradation.
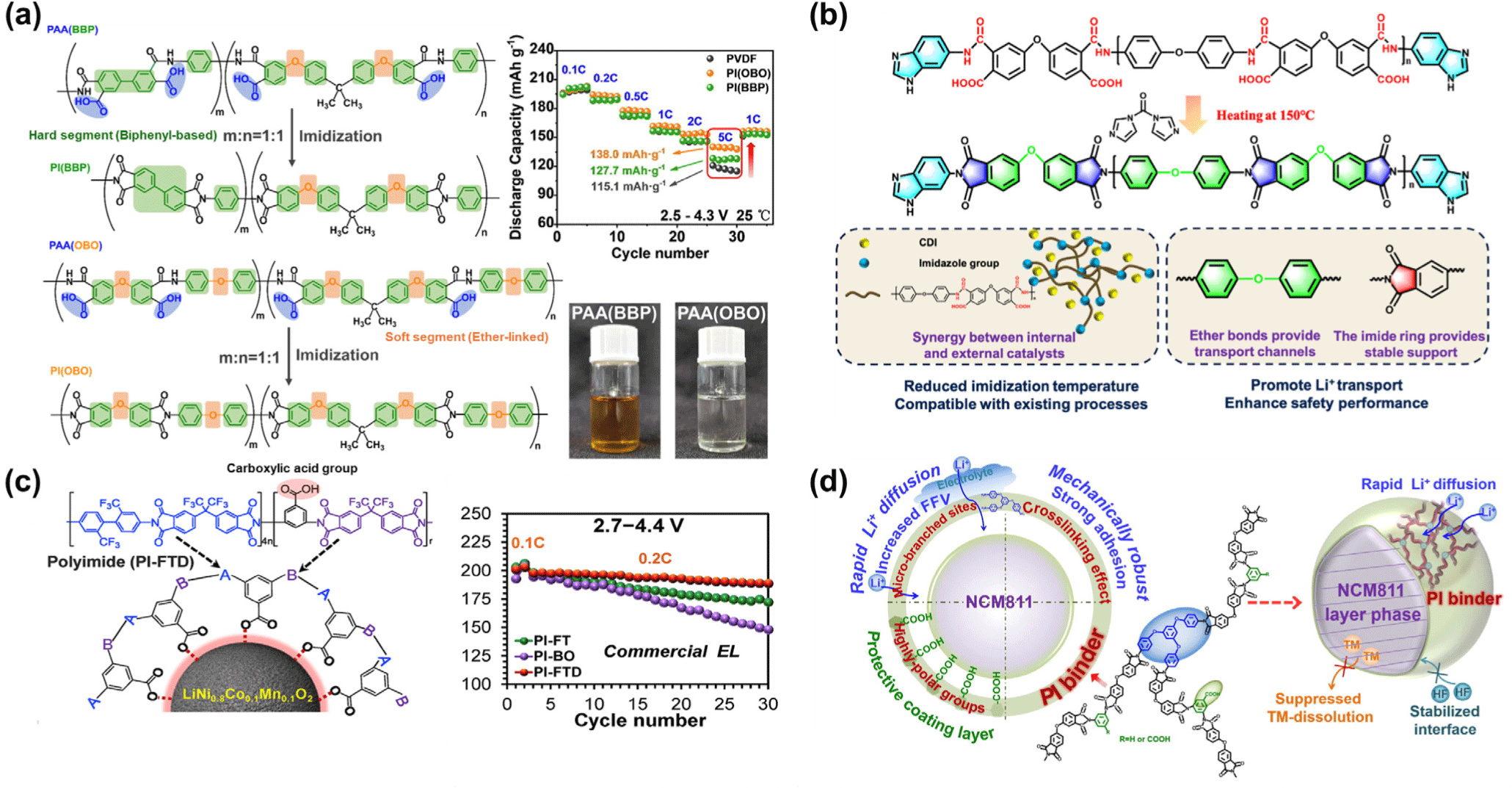
Wang et al. (2025) developed a low-temperature-imidized polyimide (LTPI) binder to overcome the limitations of conventional high-temperature processing (Figure 3b).55 By introducing ABZ (5-aminobenzimidazole) end-capping and using CDI (N,N′-carbonyldiimidazole) as a catalyst, complete imidization was achieved at 150°C. The LTPI binder enabled 84.9% capacity retention after 100 cycles at 4.3 V and delivered a discharge capacity of 119.3 mAh g−1 at 5 C (Figure 3d). As a result, the LTPI binder exhibited greatly enhanced structural and thermal stability during the charge/discharge process.
Other researchers have also explored advanced polyimide binders to enhance the electrochemical and structural stability of high-voltage NCM electrodes. Pham et al. (2019) designed a fluorinated polyimide (PI-FTD) binder that formed a strongly bonded interfacial layer on the NCM811 surface, improving oxidation resistance, interfacial stability, and flame retardancy up to 4.4 V (Figure 3c).53 Similarly, Xu et al. (2023) reported a micro-branched and crosslinked polyimide binder (PI-OmDT) incorporating –COOH groups, which effectively suppressed transition-metal dissolution and delivered 72.5% capacity retention at 4.7 V while maintaining enhanced thermal stability (Figure 3d). 54
Collectively, these studies demonstrate that polyimide binders featuring polar functional groups and crosslinked architecture can effectively stabilize high-voltage NCM cathodes and improve both cycling durability and safety.
Polyimide Binder for Anodes
Graphite is the most widely used anode material in lithium-ion batteries (LIBs), offering a theoretical capacity of 372 mAh g−1 with high energy efficiency and excellent structural stability.56,57 However, its limited capacity makes it difficult to meet the increasing energy demands required for next-generation applications. Consequently, silicon (Si) has emerged as a promising next-generation anode material due to its exceptionally high theoretical capacity of around 4200 mAh g−1.58,59 However, silicon undergoes more than 300% volume expansion during repeated lithiation and delithiation, leading to electrode cracking and unstable formation of the solid electrolyte interphase (SEI).60-63 Therefore, to enhance the performance of Si-based electrodes, it is crucial not only to improve the structural stability of the active material itself but also to optimize the binder, which maintains the integrity of the electrode and ensures continuous electronic and ionic pathways.64
In this section, the structural design strategies of polyimide binders and their mechanisms for enhancing electrochemical performance are systematically discussed, focusing on Si/C composite, SiOx, and pure Si electrodes.
Si/C composite electrodes combine the high capacity of silicon with the structural stability of graphite, forming high energy density anodes.65 However, during charge/discharge cycling, the large volume expansion of silicon induces particle separation and increased interfacial resistance.66 To mitigate these issues, incorporating polyimide (PI) binders to enhance adhesion and maintain structural integrity has proven to be an effective strategy.67
Oh et al. (2017) introduced a highly adhesive copolyimide (P84) binder to enhance the electrochemical and mechanical performance of Si/graphite (Si/C) composite anodes (Figure 4).68 The P84-based electrodes exhibited superior cycling stability and rate capability compared to PVdF-based ones, maintaining a discharge capacity of 547 mAh g−1 after 100 cycles with 93% capacity retention (Figure 4a). SEM analysis revealed that the P84-based electrodes showed significantly fewer surface cracks and morphological changes after cycling, while the PVdF-based counterparts suffered from severe cracking and delamination (Figure 4b). This enhanced structural integrity was attributed to the strong interfacial and interlayer adhesion of the P84 binder, which effectively accommodated the mechanical stresses induced by silicon volume expansion and maintained stable electrical contact during repeated lithiation/delithiation.
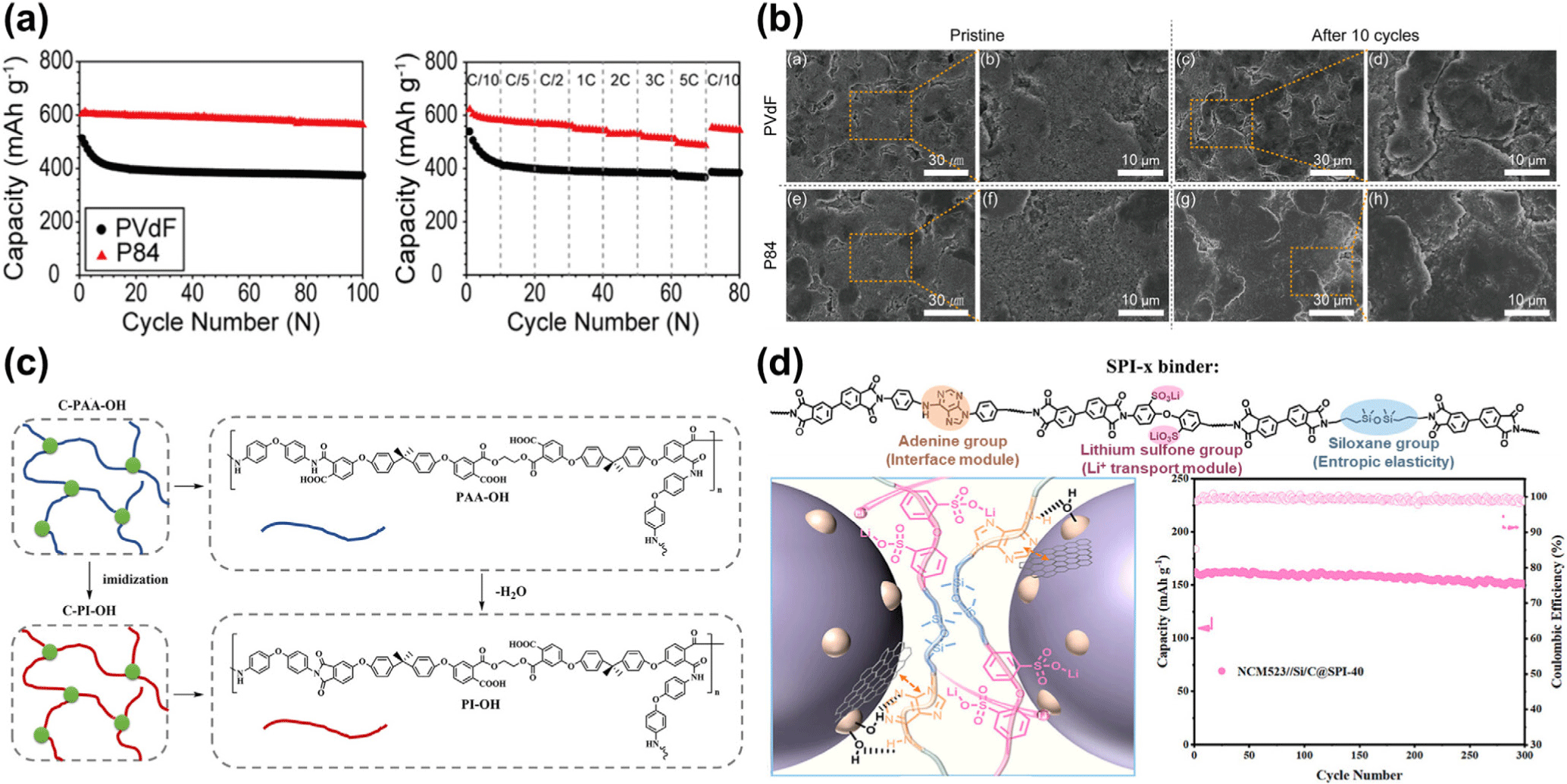
Feng et al. (2022) developed a three-dimensional cross-linked flexible polyimide (C–PI–OH) binder incorporating ethylene glycol (EG) and 1,3,5-benzenetricarboxylic acid (H3BTC) (Figure 4c).69 The C–PI–OH binder formed a covalent and hydrogen-bonding network with silicon and the current collector through its free carboxyl (–COOH) and amide (–CONH–) groups, while its flexible polymer chains effectively accommodated the expansion and contraction of silicon during cycling. As a result, the electrode maintained a capacity of 1112 mAh g−1 after 200 cycles, exhibiting superior adhesion strength, ionic conductivity, and cycling stability compared to PVDF and rigid PI binders.
In addition, various multifunctional polyimide binders are being actively studied to simultaneously improve the mechanical strength, ion/electron conductivity, and interfacial stability of Si/C electrodes. Tan et al. (2025) developed an aqueous copolyimide (SPI-x) binder consisting of three modules of adenine, lithium sulfone group, and siloxane to realize double interaction with both Si and C surfaces and excellent lithium-ion transport properties (Figure 4d).70 Similarly, Yang et al. (2025) synthesized fluorine-containing soluble polyimides (PI-FS, PI-FN), introducing siloxane and amine groups to improve adhesion, conductivity, and long-term cycle stability.71
Collectively, these studies demonstrate that the rational molecular engineering of polyimide binders can balance mechanical strength, ion/electron transport, and interfacial stability, ensuring durable performance of Si/C composite anodes.
SiOx electrodes suffer from limited long-term cycle life due to the structural heterogeneity of Si–O bonds and the unstable SEI layer formed during repeated lithiation/ delithiation.72 Therefore, it is essential to design binders capable of forming stable Si–O–C bonds at the interface to improve chemical stability.73
Kang et al. (2024) developed a high-toughness polyimide (PDMI) binder with a rigid–soft hybrid structure by copolymerizing 4,4′-oxydiphthalic anhydride (ODPA), 4,4′-oxydianiline (ODA), 3,5-diaminobenzoic acid (DABA), and 1,3-bis(3-aminopropyl)-tetramethyl disiloxane (DMS) (Figure 5a).77 The aromatic imide backbone provided high mechanical strength (≈150 MPa), while the siloxane (Si–O– Si) chains imparted flexibility and facilitated lithium-ion transport, increasing the lithium-ion diffusion coefficient (Dli+) from 9.8 × 10−1 to 1.6 × 10−9 cm2 s−1. Moreover, the strong binding affinity of the carboxyl (–COOH) groups formed stable chemical bonds with the SiOx surface, resulting in 85 % capacity retention after 500 cycles at 1.3 A g−1. Overall, the PDMI binder effectively suppressed electrode expansion, stabilized the SEI, and reduced interfacial resistance, thereby significantly improving the long-term stability of high-capacity SiOx anodes.
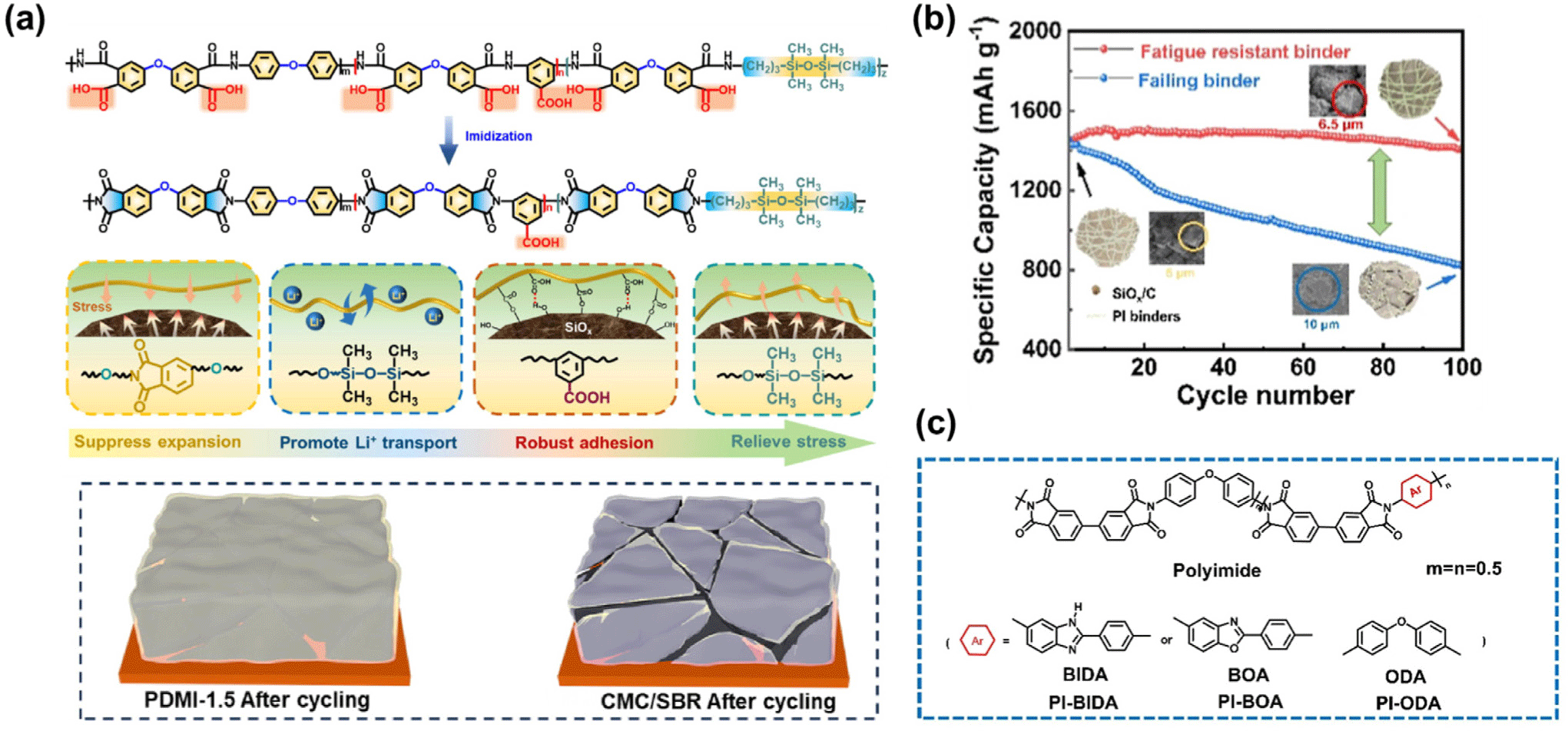
In addition to pure SiOx systems, recent studies have extended the application of polyimide binders to SiOx/C composite anodes, where the carbon matrix enhances electrical conductivity and buffers volume change. Li et al. (2025) developed fatigue-resistant BPDA-based PI binders (BPDA-ODA and BPDA-PDA) that maintained over 95 % capacity retention after 100 cycles by effectively accommodating repeated stress and suppressing SiOx/C electrode swelling (Figure 5b).75 Subsequently, Li et al. (2025) reported a benzimidazole-containing dual-function PI (PI-BIDA) binder that strengthened Li+ affinity and stabilized a LiF-rich SEI layer, achieving 90 % retention after 150 cycles and enhanced interfacial stability (Figure 5c).76
Polyimide binders effectively enhance the interfacial stability of SiOx and SiOx/C anodes by promoting strong chemical bonding, accommodating volume changes, and improving Li+ transport, thereby achieving outstanding cycling durability and SEI stability.
Pure silicon electrodes are highly prone to structural failure due to volume expansion during cycling.78 Therefore, polyimide (PI) binders possessing both mechanical strength and flexibility play a crucial role in maintaining electrode integrity.
Choi et al. (2015) developed a high-adhesion copolyimide (P84) binder by copolymerizing 3,3′,4,4′-benzophenonetetrac arboxylic dianhydride (BTDA), toluene diisocyanate (TDI), and methylene diphenyl diisocyanate (MDI).79 The combination of an aromatic imide backbone and a benzophenone structure provided high tensile strength while maintaining solubility in organic solvents such as NMP and DMF, allowing uniform electrode formation without high-temperature imidization. The P84 binder exhibited strong chemical bonding with the silicon surface, improving interfacial adhesion and mechanical durability (Figure 6a). Consequently, the electrode delivered an initial capacity of 2392 mAh g−1 and retained 1313 mAh g−1 after 300 cycles at 1.2 A g−1, demonstrating excellent cycling stability.
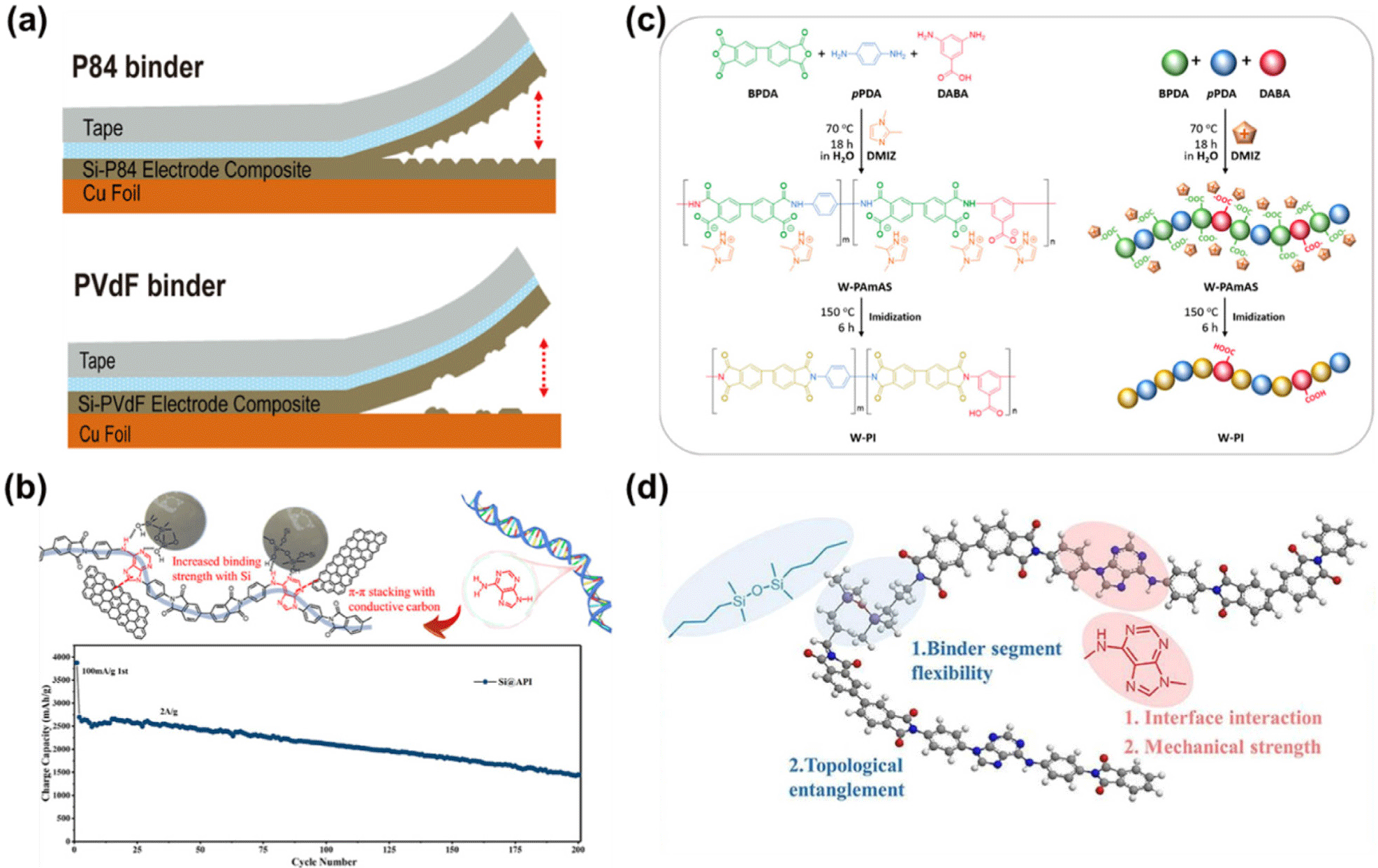
Tan et al. (2023) designed an adenine-derived polyimide (API) binder synthesized from 3,3′,4,4′-biphenyltetracarboxy lic dianhydride (BPDA) and 9,10-bis(4-aminophenyl)adenine (p-APA).80 The nitrogen-rich adenine rings and aromatic π-electron structure enabled simultaneous π–π interactions and hydrogen bonding with silicon particles and conductive carbon, constructing a dual-interaction network within the electrode. This structural design mitigated silicon volume expansion and enhanced interfacial stability, resulting in an initial discharge capacity of 2163 mAh g−1 and 81.2% capacity retention after 100 cycles at 0.5 C (Figure 6b).
So et al. (2021) synthesized a water-processable poly(amic acid) salt (W-PAmAS) precursor via an aqueous reaction using 3,5-diaminobenzoic acid (DABA) and pyromellitic dianhydride (PMDA) with 1,2-dimethylimidazole (DMIZ) as a catalyst (Figure 6c).81 The precursor was then converted into a water-based polyimide (W-PI) binder through low-temperature imidization at 150°C. The –COOH groups of DABA reacted with surface –OH groups on Si to form ester bonds (Si–O–C), strengthening interfacial adhesion while achieving both environmental friendliness and a low manufacturing temperature. As a result, the W-PI binder electrode exhibited an initial capacity of 2061 mAh g−1 and retained 1883 mAh g−1 after 200 cycles at 1.2 A g−1, showing outstanding stability and more than twice the adhesion strength of PVDF, thereby significantly enhancing both mechanical and electrochemical durability.
In recent studies, Tan et al. (2024) developed a rigid– flexible copolyimide (API-x) binder that integrates adenine-based rigid segments and siloxane–alkane flexible chains, achieving a balanced combination of mechanical strength and flexibility.82 This structural design effectively accommodates the large volume expansion of silicon and enhances long-term electrode stability (Figure 6d).
These studies demonstrate that rational design of polyimide binders with balanced rigidity and flexibility effectively improves the mechanical and interfacial stability of pure Si anodes.
Conclusion and Outlook
In next-generation high-capacity lithium-ion battery electrodes, significant structural and volumetric changes occurring during charge–discharge cycles undermine the stability of conventional binder systems. To overcome these challenges, polyimide-based binders incorporating various functional groups have emerged as promising next-generation materials. Polyimides exhibit excellent thermal and mechanical stability derived from the resonance stabilization of the imide ring and the aromatic backbone. Moreover, the incorporation of functional groups such as carboxyl (–COOH), amine (–NH2), ether (–O–), and sulfonic acid (–SO3H), along with controlled crosslinking, enables a balanced enhancement of interfacial adhesion, ionic conductivity, and structural flexibility.83,84
Accordingly, the development of multifunctional polyimide binders through precise molecular-level structural design has gained increasing attention. By carefully combining rigid and flexible molecular segments and employing various noncovalent interactions, such as hydrogen bonding, π–π stacking, and ionic interactions, both interparticle cohesion and charge transport pathways within the electrode can be simultaneously strengthened.
In particular, water-based synthesis strategies have been recognized as highly promising approaches for industrial scalability, as they minimize the use of organic solvents while preserving the intrinsic thermal stability and adhesive properties of polyimides.
Future research on polyimide binders should advance toward rational design approaches that simultaneously achieve high energy density, long cycle life, and environmental sustainability. Three major research directions are proposed:
-
(1) Development of redox-stable polyimide frameworks through copolymerization with conductive polymers.
-
(2) Establishment of sustainable synthesis processes, including aqueous polyimide synthesis and bio-based monomer design.
-
(3) Implementation of cost-effective and scalable strategies for mass production and commercialization.
Furthermore, polyimide binders incorporating function-alities such as self-healing capability, mechanical flexibility, and environmental responsiveness are expected to enable an expanded application scope. This perspective is further supported by recent studies on mechanically adaptive polymer systems that maintain electrical performance and structural integrity under substantial deformation, highlighting the broader applicability of multifunctional polymer design principles beyond conventional applications.85
In conclusion, the integration of sophisticated molecular design and water-based synthesis in polyimide-based binders represents a key strategy for achieving both eco-friendliness and high performance, positioning this approach as a promising technology to accelerate the commercialization of next-generation high–energy-density batteries.






