Introduction
Anhydrous liquid ammonia is widely used in the chemical industry, refrigeration systems, energy storage, and transportation sectors.5,14,15 However, due to its high reactivity and penetrability, it can cause severe corrosion issues in both metallic and non-metallic materials.1,2,3,6,9,13 Especially when anhydrous liquid ammonia is stored and transported, rubber-based sealing and packaging materials could experience their lifespan reduction with extended exposure, which would ultimately cause safety concern.11
EPDM (Ethylene Propylene Diene Monomer) is widely used in seals, gaskets, and other applications due to its excellent ozone resistance, heat resistance, oxidation resistance, and insulation properties.12 However, in extreme environments such as exposure to anhydrous ammonia, it is difficult to ensure adequate stability when used alone. Therefore, silica, which has excellent dispersibility, mechanical reinforcing effects, and chemical stability, has attracted attention as an inorganic reinforcing agent.8 Various inorganic fillers have been examined to improve the interfacial adhesion between EPDM and the matrix, but silica stands out because it can provide both mechanical reinforcement and chemical stability simultaneously.
However, since EPDM is inherently a non-polar rubber, direct interfacial interactions with the polar hydroxyl groups (-OH) on the silica surface are limited, which can lead to problems such as filler aggregation or poor dispersibility in EPDM-silica elastomer composites. Although the hydroxyl groups on the silica surface are difficult to be covalently linked with EPDM, they act as reactive sites that can enhance interfacial affinity through silane coupling agents or surface modification. This, in turn, plays a bridging role between EPDM and silica, contributing to the enhancement of composite performance.
While numerous studies have been conducted to improve the chemical resistance of EPDM, research on its stability in highly corrosive and specialized environments, such as 99.5% anhydrous liquid ammonia, is very limited.4,13 In particular, anhydrous ammonia has both strong polarity and high penetrability, which causes chemical degradation and mechanical performance deterioration to occur rapidly when EPDM is used alone. This leads to a lack of foundational data for practical sealing material and gasket design, resulting in difficulties in material selection in industrial applications.
The aim of this study is to evaluate the chemical performance of EPDM elastomer composites filled with precipitated silica in 99.5% anhydrous liquid ammonia environment. Through this, the study seeks to address the limitations of using EPDM alone and identify the effects of silica filling on chemical resistance and mechanical stability. Specifically, by analyzing the changes in various properties due to silica reinforcement, this study aims to clarify the influence of the EPDM-silica interfacial interaction and provide foundational data for the development of high-performance sealing materials that can be applied in extreme chemical environments.
Experimental
In this study, EPDM was used as the matrix, specifically Kumho Polychem’s KEP 570F. This material has an Ethylene content of 70±3 wt% and an ENB (Ethylidene Norbornene) content of 4.5±1.0 wt%, which provides excellent crosslinking reactivity and chemical resistance. For enhancing chemical resistance and reinforcement, Precipitated Silica (PS) was used as the filler. Four types of silica with different SSA were applied: ULTRASIL 360 (SSA: 55 m2/g), ULTRASIL VN2 (SSA: 130 m2/g), ULTRASIL 7000GR (SSA: 175 m2/g), and ULTRASIL 9100GR (SSA: 235 m2/g). The silica content was maintained at 20 phr, and Si-69 (bis(triethoxysilylpropyl) tetrasulfide) was used at 1.6 phr(parts per hundred rubber) as a silane coupling agent to improve the bonding between the rubber and silica. Additionally, Polyethylene Glycol 4000(PEG-4000) was added at 1.6 phr as a dispersing aid. The crosslinking system used was a peroxide-based system, with Dicumyl Peroxide (DCP) as the crosslinking agent at 1.5 phr. Zinc Oxide (ZnO) and Stearic Acid were used as processing and dispersion aids, at 5 phr and 1 phr, respectively.
The EPDM-silica elastomer composites were prepared using a 300cc internal mixer (Anton Paar, Austria) with a tangential rotor, an 8-inch open roll mill (Pungkwang Machinery, Korea), and a Hot press (SJHT120, SMDMC Co., Ltd., Korea). The processing conditions for the internal mixer were set to a fill factor of 0.78 and 50 rpm. The EPDM was preheated for 1 minute to enhance its plasticity, and then additives (ZnO and Stearic acid) were added. After mixing for 30 seconds, silica, Si-69, and PEG 4000 were added in three separate portions to prepare the Masterbatch (MB). Next, DCP was added at 1.5 phr to produce the Final Masterbatch (FMB), and mixing was completed within 1 minute to prevent scorching. The composition of the EPDM elastomer composite prepared in this study is shown in Table 1. To observe the effect of silica SSA on the EPDM elastomer composite, four types of silica with SSAs were added and evaluated.
3. Experimental Methods
The processability of the EPDM-silica elastomer composite samples was evaluated using a Mooney viscometer (DMV-200C, Daekyung, Korea). The samples were preheated at 100°C for 1 minute and then tested for 4 minutes, with the torque value measured on the viscometer (ML(1+4) 100°C).
The crosslinking behavior of the EPDM-silica elastomer composite samples was evaluated using a Moving Die Rheometer (Nichigo Shoji Co., Ltd., Japan) at a temperature of 170°C based on the standard, JIS K 6300-2. The torque values applied to the samples, scorch time (t10), and optimum curing time (t90) were measured.
EPDM-silica elastomer composite samples were immersed in 99.5% anhydrous liquid ammonia tank for 168 hours. Considering that the liquefaction temperature of ammonia is −33°C, the immersion tests were conducted at (−35 ±1)°C.
The crosslinking density of the EPDM-silica elastomer composite samples before and after 99.5% anhydrous liquid ammonia immersion was measured based on ASTM D6814. Using the weight and swelling ratio of the samples, the crosslinking density was calculated by applying the Flory-Rehner equation.11
v = Crosslinking density [mol/g]
MC = Average molecular weight between crosslink points [g/mol]
V1 = Volume fraction of rubber in the swollen gel at equilibrium
V0 = The molar volume of the solvent [cm3/mol]
ρr = The density of the rubber sample [g/cm3]
χ = Interaction parameter between the rubber and solvent
β1 = 0.34
δp = Solubility parameter of the polymer
δS = Solubility parameter of the solvent
The volume change ratio of EPDM-silica elastomer composite samples after immersion in 99.5% anhydrous liquid ammonia was analyzed based on the KS M 6518 immersion test standard. Test specimens with dimensions of 20 mm in width, 50 mm in length, and (2.00±0.15) mm in thickness were prepared in sets of five and immersed in 99.5% anhydrous liquid ammonia for 168 hours. The density and weight of the samples were measured before and after immersion to calculate the volume and volume change ratio. The average value, excluding the maximum and minimum values, was used for the analysis.
The surface structure of the EPDM-silica elastomer composite samples before and after 99.5% anhydrous liquid ammonia immersion was analyzed using FE-SEM (Field Emission Scanning Electron Microscope, JEOL, Model: JSM-6701F) and AFM (Atomic Force Microscope, Oxford Instruments, Model: Cypher S).7 In the FE-SEM analysis, the samples were sufficiently cooled with liquid nitrogen and then fractured to observe both the fracture surface and the surface. The AFM measurement range was 10 μm × 10 μm, and the surface roughness was analyzed at three different points.
The tensile strength change ratio of EPDM-silica elastomer composite samples before and after immersion in 99.5% anhydrous liquid ammonia was analyzed based on KS M 6518. The sheet form vulcanizates were cut into dumbbell-shaped specimens (parallel part width: 5 mm, parallel part length: 20 mm, parallel part thickness: 2 mm, distance between grips: 20 mm), and the tensile strength was measured at room temperature with a speed of 500 mm/min using a Universal Testing Machine (Model: Instron 3345, Instron, USA). Tests were conducted on five samples for each condition, and the measured values were analyzed using the average, excluding the maximum and minimum values. The tensile strength change ratio was calculated using the average values of the specimens before and after ammonia immersion according to the following formula:
TS0 = Tensile strength before ammonia immersion
TS1 = Tensile strength after ammonia immersion
The dynamic mechanical properties of EPDM-silica elastomer composite samples before and after immersion in 99.5% anhydrous liquid ammonia were measured using an ARES G2 (TA Instruments, USA). The temperature-sweep test was conducted in tension mode over a temperature range of –70 to 50°C (5°C/min), at a strain of 0.1% and a frequency of 10 Hz. Glass transition temperature, storage modulus, loss modulus, and loss tangent were analyzed to compare the viscoelastic properties of the EPDM elastomer composites.
Results and Discussion
The results of Mooney viscosity measurements after blending four types of silica into EPDM are shown in Figure 1 and Table 2. The Mooney viscosity increased with the SSA of the silica increased. This indicates that as the SSA of the silica increases, the interaction with the EPDM matrix intensifies, which in turn affects the viscosity.
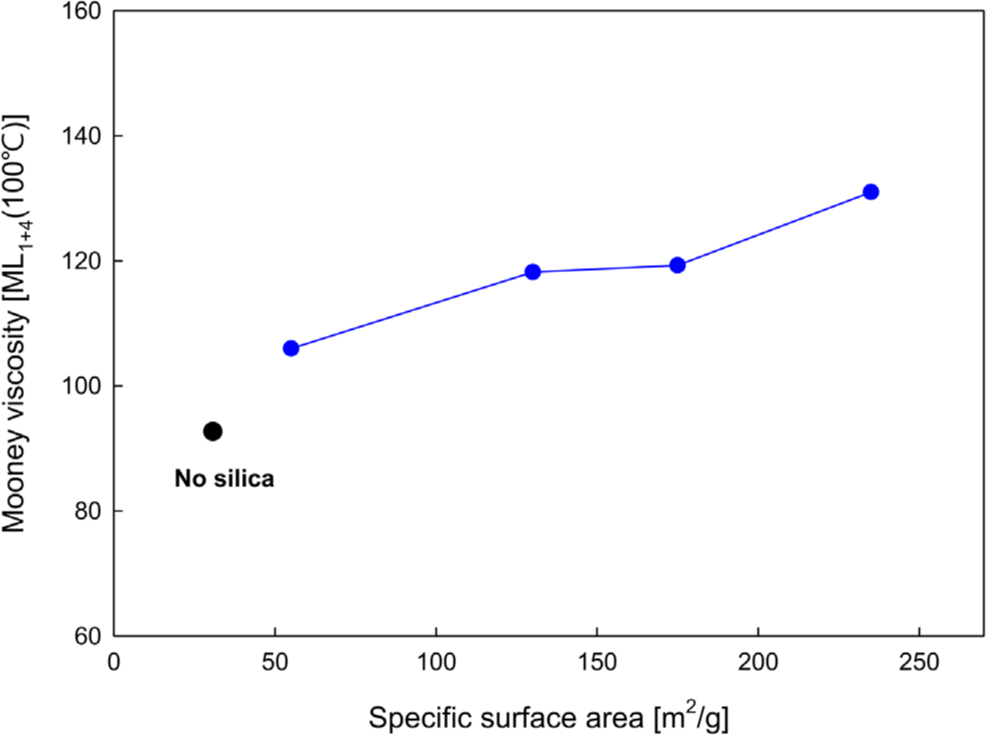
| Neat | PS_55 | PS_130 | PS_175 | PS_235 | |
|---|---|---|---|---|---|
| Mooney viscosity [ML1+4100°C] | 91.9 | 106.0 | 118.2 | 119.3 | 131.0 |
The results of crosslinking behavior after blending four types of PS into EPDM are shown in Figure 2, Figure 3, and Table 3.
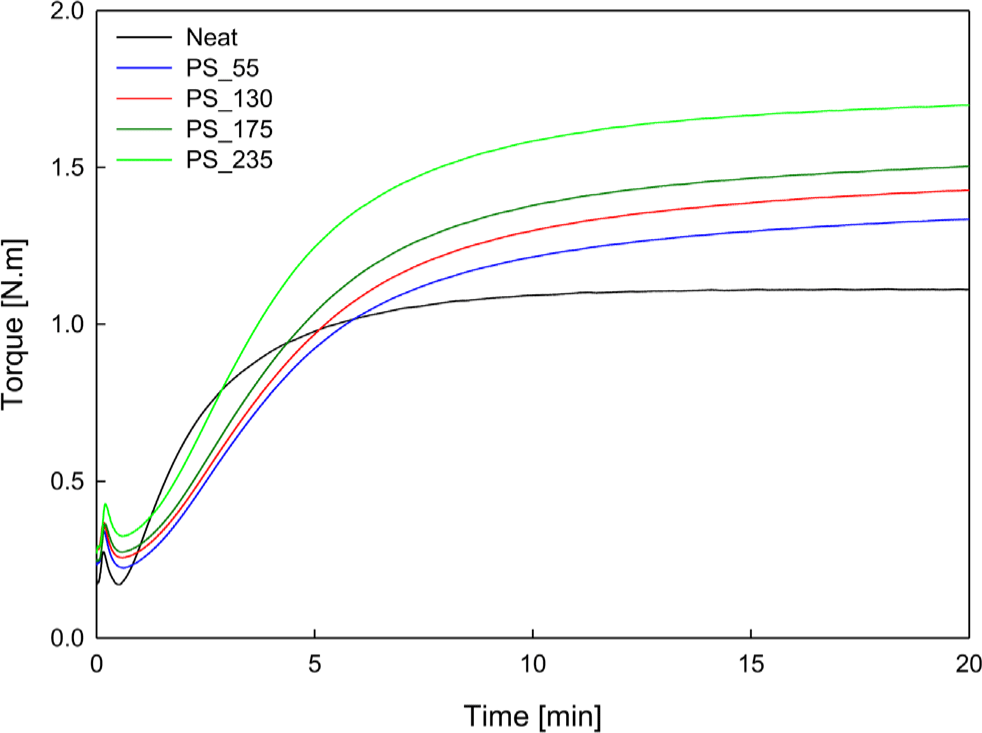
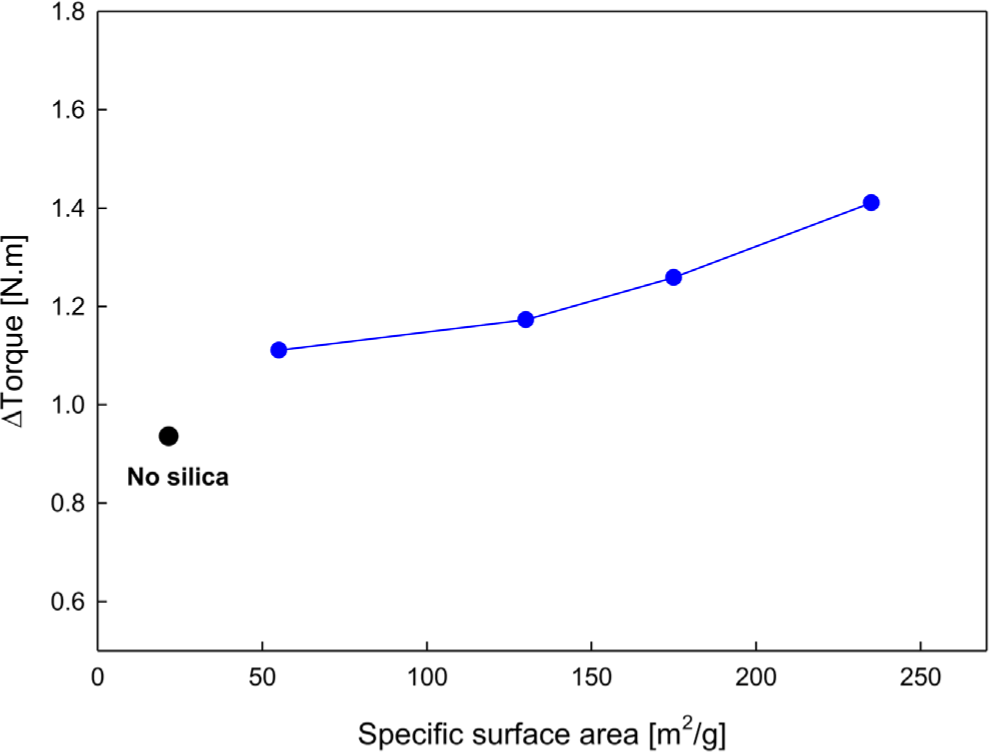
As the SSA of the silica increased, Δtorque increased, and the optimum curing time (t90) generally decreased. Although there were some deviations, the results were generally consistent. This can be interpreted as the PS surface being rich in hydroxyl groups (-OH), which enables good dispersion within the EPDM matrix and active participation in the crosslinking reaction.
The samples prepared by blending four types of silica into EPDM were immersed in 99.5% anhydrous liquid ammonia, and the properties of the samples before and after immersion were compared and analyzed.
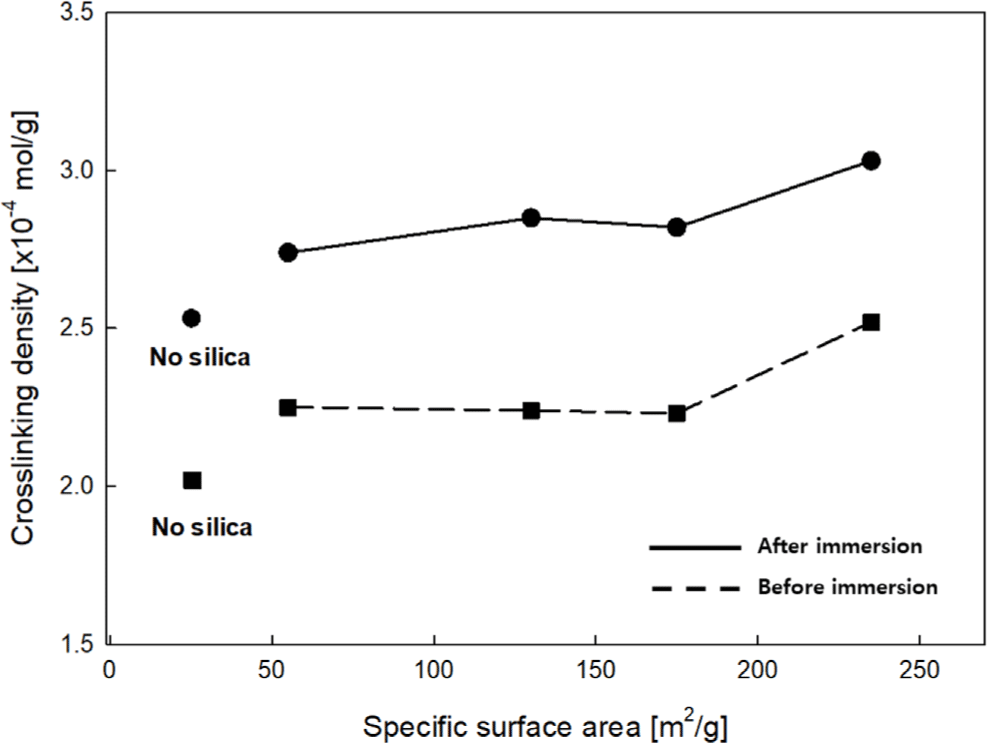
The crosslinking density of the EPDM-silica elastomer composite samples before and after immersion in 99.5% anhydrous liquid ammonia was compared and is shown in Figure 4. As the SSA of the silica increased, the crosslinking density showed an increasing trend. This can be interpreted as the increase in the interaction between EPDM and silica with a higher SSA, which leads to improved dispersion and bonding strength. These results are consistent with the trend observed in the Δtorque analysis from the crosslinking behavior analysis. Furthermore, an increase in crosslinking density was observed after immersion in 99.5% anhydrous liquid ammonia. As a result, it can be concluded that the surface structure of the silica particles changes during the immersion process, enhancing the interaction between EPDM and silica, which leads to the formation of a dense crosslinking network.
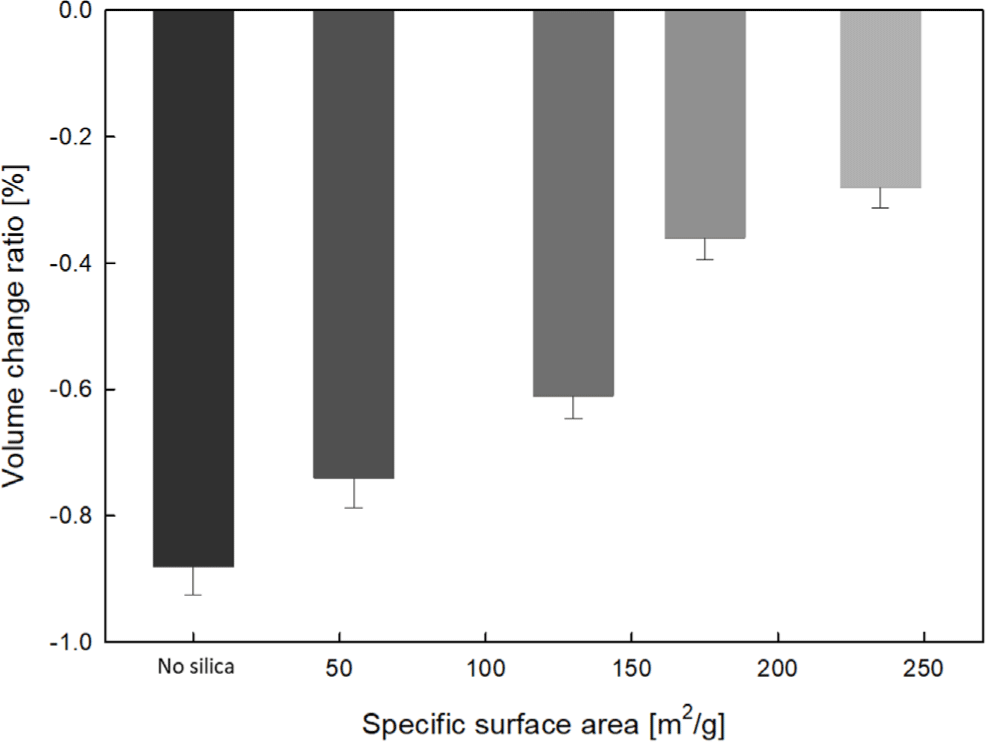
The results of the volume change ratio comparison before and after immersion of EPDM-silica elastomer composite samples in 99.5% anhydrous liquid ammonia are shown in Figure 5. Both the neat (no silica) samples and the silica-filled samples showed a decrease in volume. This can be understood as ammonia penetrating the EPDM matrix during the immersion process, inducing contraction of the polymer chains and structural rearrangement. The volume reduction ratio was observed to decrease with increasing SSA of silica.
The results of the tensile strength change ratio comparison before and after immersion of EPDM-silica elastomer composite samples in 99.5% anhydrous liquid ammonia are shown in Figure 6. After immersion, the tensile strength generally decreased, and the tensile strength change ratio decreased as the SSA of the silica increased. This trend is similar to the volume change ratio results presented earlier and is attributed to ammonia penetrating the EPDM matrix during the immersion process, inducing contraction of the polymer chains and structural rearrangement.
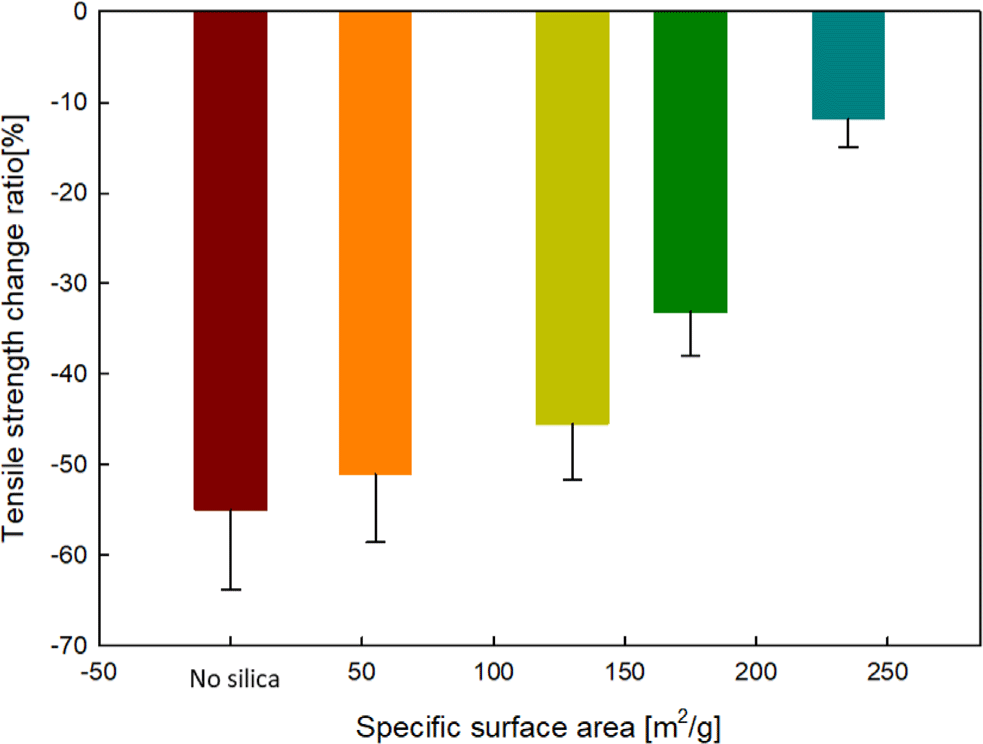
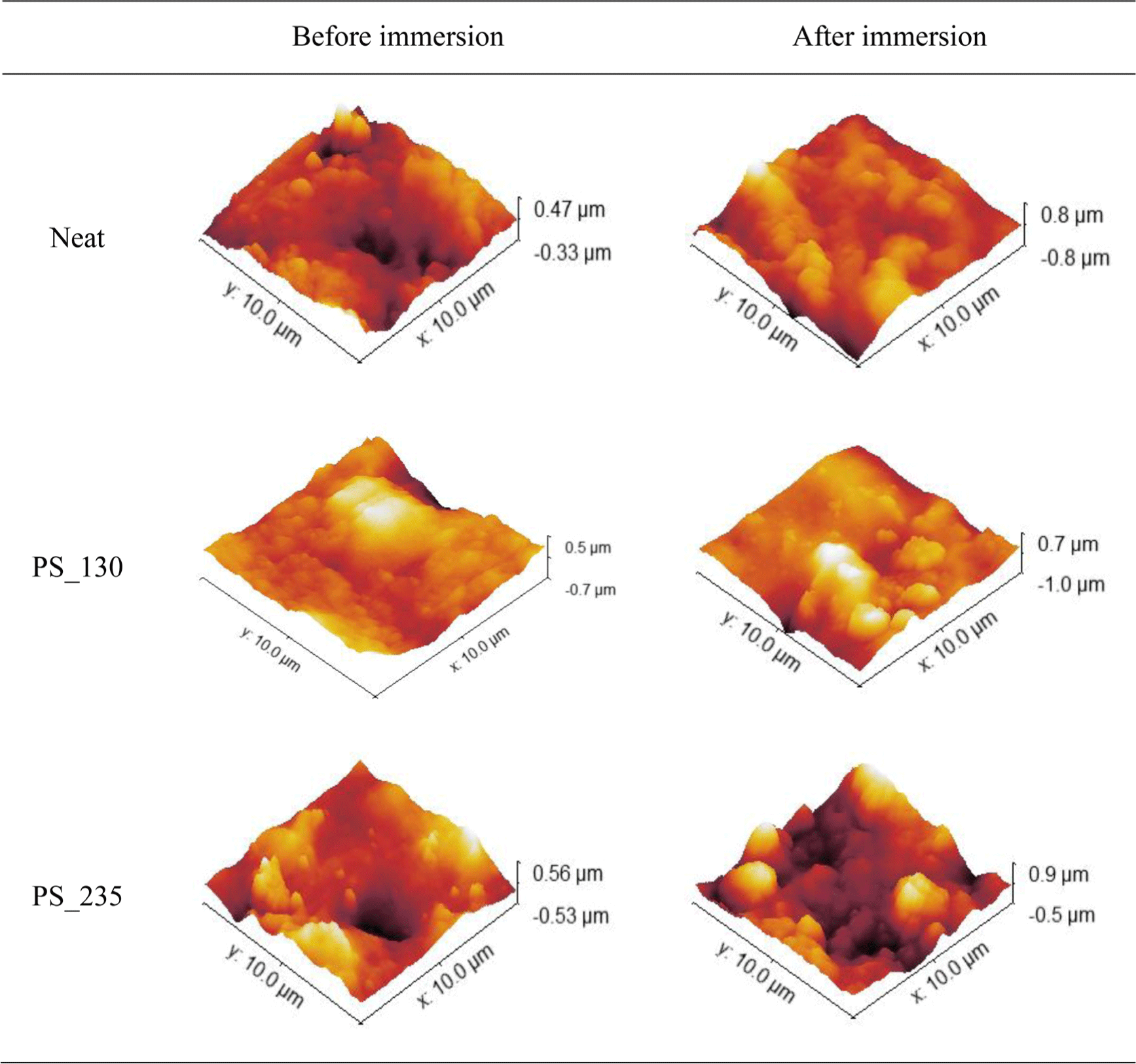
To compare the morphology before and after immersion in 99.5% anhydrous liquid ammonia, neat samples and PS_130, PS_235 samples were selected for analysis. The morphology of these samples was analyzed using FE-SEM, and the results are shown in Figure 7 and Figure 8.


The fracture surface morphology, excluding the geometric patterns formed during sample fracture, was similar before and after immersion. In contrast, the surface morphology showed an increase in roughness after ammonia immersion. This increase was more pronounced in PS_130 with smaller silica SSA compared to PS_235.
Additionally, the surface roughness values were analyzed using AFM, and the results are presented in Figure 9 and Table 4. The comparison of roughness parameters (Ra, Rq, Rz) revealed that the increase in these values confirmed the enhancement of surface roughness after ammonia immersion. This trend was also more pronounced in PS_130 compared to PS_235. This result confirms the observations mentioned earlier in the volume change ratio and tensile strength change ratio. Therefore, it is concluded that the chemical resistance to ammonia increases as the SSA of silica increases. Furthermore, under the condition of 168 hours of immersion in anhydrous liquid ammonia, the surface roughness increased, but no effect was observed on the cross-section. Thus, under the 168-hour immersion condition, it was confirmed that the surface changes of the EPDM-silica elastomer composite were more significant than those in the fracture surface.
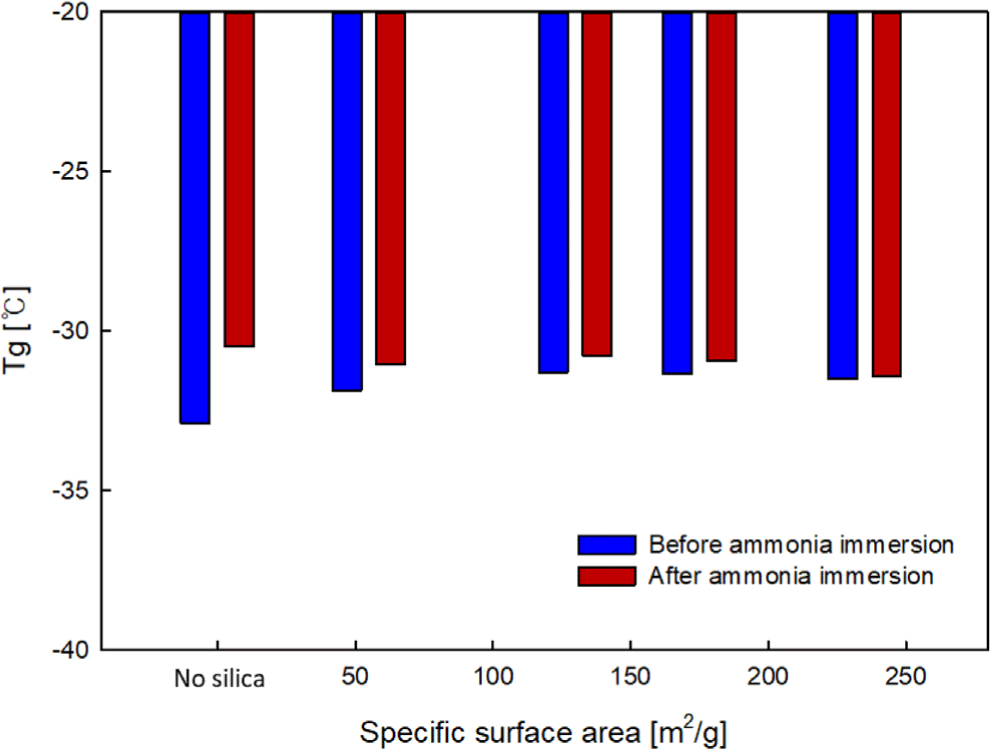
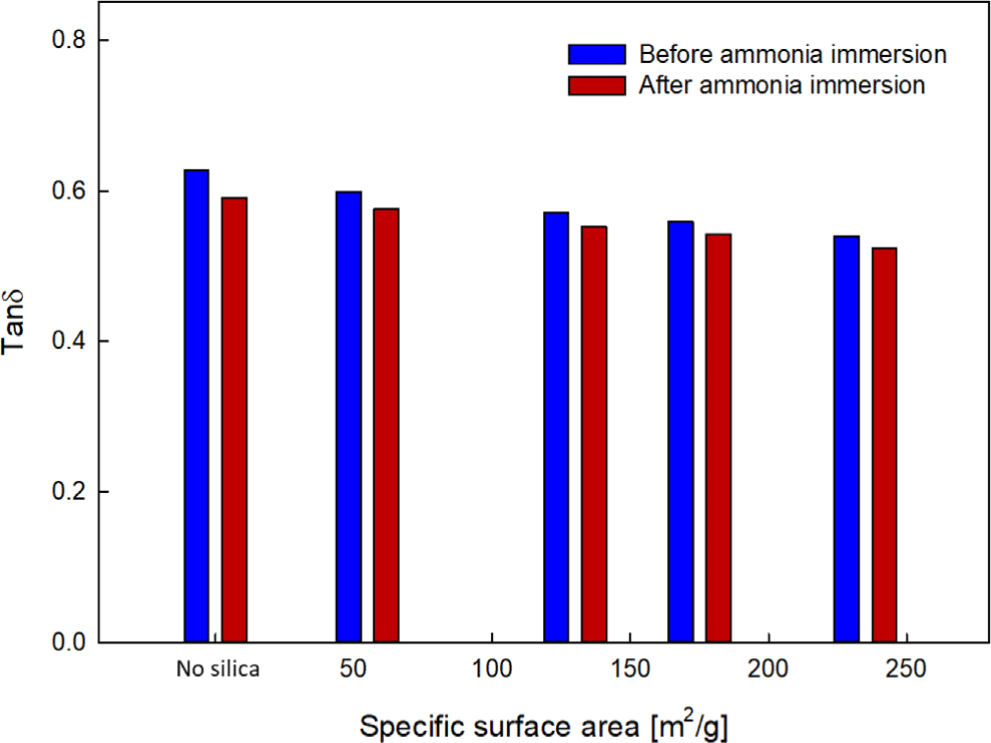
After immersing the EPDM-silica elastomer composite samples in 99.5% anhydrous liquid ammonia, the dynamic mechanical properties of the samples before and after immersion were analyzed. The glass transition temperature (Tg) and loss tangent (tanδ) before and after immersion are shown in Figure 10, Figure 11, and Table 5. After immersion in 99.5% anhydrous liquid ammonia, an increase in Tg and a decrease in tanδ were observed. This result can be attributed to the increased crosslinking density, which restricts the free movement of the polymer chains. Neat EPDM samples, which have greater chain freedom, showed the highest increase in Tg and a decrease in tanδ among the prepared systems after immersion in 99.5% anhydrous liquid Ammonia. In contrast, the silica-filled samples, resulted in a limited increase in Tg and a decrease in tanδ after immersion, which might be caused by the restriction of the EPDM chains due to the addition of PS. This trend was found to be more pronounced as the specific surface area (SSA) of silica increased.
Conclusions
In this study, the properties of EPDM-silica elastomer composites with four types of silica having different SSAs were analyzed before and after immersion in 99.5% anhydrous liquid ammonia.
-
Processability and Crosslinking Behavior: As the SSA of silica increased, the Mooney viscosity and ΔTorque increased, while the optimum curing time (t90) gradually decreased.
-
Crosslinking Density and Volume Change Ratio: After ammonia immersion, the crosslinking density increased in all compositions, and the volume change decreased in all samples. The reduction in volume change was smaller as the SSA increased.
-
Mechanical Strength: The tensile strength decreased after immersion, but the decrease in tensile strength was less pronounced as the SSA increased.
-
Morphology/Surface Roughness: No distinct structural changes were observed in the fracture surface morphology. However, the surface roughness increased after immersion, and the magnitude of the increase became smaller as the SSA of the silica increased.
-
Dynamic Mechanical Behavior: After immersion in ammonia, a common increase in Tg and a decrease in tanδ were observed. The change was most significant for Neat samples, and the change became smaller as the SSA increased.
Therefore, this study demonstrated that the use of silica with a larger SSA resulted in smaller property changes after immersion, indicating improved chemical stability of the EPDM–silica elastomer composites. This highlights the importance of selecting silica SSA as a key design factor when designing sealing and gasket materials for use in liquid ammonia environments.4,8,11,12
