Introduction
Rubber vulcanizates are widely used in various applications such as automotive components, sealing materials, and footwear due to their excellent elasticity, vibration absorption, and sealing properties. These materials possess the ability to recover their original shape after deformation through the crosslinked polymer network formed during vulcanization. However, when subjected to prolonged thermal or mechanical stress, rubber vulcanizates may undergo permanent deformation, which can critically impair their functionality in service environments.
One of the primary causes of permanent deformation is a change in the crosslink density.1 Thermal aging promotes the formation of additional crosslinks, resulting in increased rigidity and suppression of elastic recovery.2,3 If vulcanized rubber undergoes deformation under these conditions, the restored shape may deviate significantly from its original form. Nevertheless, when the molecular structure (such as crosslink density, crosslink type, and polymer chain alignment) is maintained within the elastic limit, the material can exhibit full recovery without permanent set.
To enhance the thermal and mechanical stability of rubber compounds, thermoplastic materials have been increasingly explored as blending agents.2,3 Thermoplastic resins, unlike thermosetting rubbers, can be repeatedly softened and remolded upon heating. While their heat resistance is generally inferior, they offer processability advantages and tunable mechanical properties by adjusting plasticizer content.
Recent studies have shown that blends of thermoplastic polyurethane (TPU) with rubber demonstrate improved abrasion resistance and elastic recovery.4,5 Additionally, composites of thermoplastic olefin (TPO) with natural rubber have been reported to provide a balance of excellent processability and mechanical properties.6 Blending rubber with thermoplastic resins, particularly thermoplastic elastomers, has emerged as an effective strategy to balance elastic recovery and mechanical durability.
In this study, rubber/thermoplastic composites were prepared using a blend of vulcanizable rubber, thermoplastic resin (PVC), and plasticizers. The formulations varied in terms of plasticizer and vulcanization accelerator content to assess their influence on shape recovery behavior. The recovery performance was evaluated using a circular deformation test at both room temperature and elevated temperature (80°C), providing insight into the effect of thermoplastic blending on the elastic behavior and thermal aging resistance of rubberbased materials.
The rubber/thermoplastic composite developed in this study exhibits significantly improved mechanical strength and abrasion resistance, making it suitable for applications such as footwear outsoles, industrial cushioning materials, and durable consumer or sports goods that experience repetitive loading under low- to mid-temperature conditions. However, due to the thermoplastic phase, its shape-recovery performance decreases at elevated temperatures, limiting its use in components that require long-term thermal stability, such as automotive engine-bay parts or high-heat industrial equipment.
Experimental
To evaluate the shape recovery behavior of rubber/ thermoplastic composites, a range of polymeric materials and additives were utilized. Natural rubber (NR, Vietnam) was employed as the base elastomer due to its vulcanization capability and elastic properties. Polyvinyl chloride (PVC, Hanwha Solutions) was selected as the thermoplastic resin, and dioctyl phthalate (DOP) was used as a plasticizer to improve the processability and flexibility of the PVC phase. Sulfur and two types of vulcanization accelerators (Accelerator A and Accelerator B) were incorporated into the formulation to promote crosslink formation. Sulfur served as the primary curing agent, while Accelerator A facilitated rapid initiation of vulcanization and Accelerator B contributed to crosslink stability during the later stages of curing. Filler was added as a reinforcing filler, and a silane coupling agent was used to enhance dispersibility between the rubber and filler phases.
Rubber/thermoplastic composite specimens were prepared using conventional melt-mixing and compression molding techniques. The raw materials, including natural rubber, PVC, DOP, sulfur, vulcanization accelerators, fillers, and coupling agents, were weighed according to the formulations listed in Table 1. The blending process was performed using a laboratory-scale internal mixer (kneader) to ensure homogeneous dispersion of thermoplastic resin and additives within the rubber matrix.
Following preliminary mixing, the compound was further processed using a two-roll mill to improve sheet uniformity and remove trapped air. The resulting sheets were then cut into appropriate sizes for vulcanization.
Vulcanization was carried out using a hot press at 155°C, with curing times determined by cure curves obtained from an oscillating disc rheometer (ODR). The optimum curing time (t90) was used as the standard vulcanization condition for all formulations.
For mechanical and recovery tests, the vulcanized sheets were die-cut into rectangular strips with dimensions suitable for circular deformation testing, as specified by relevant standards. Specimens were stored in a desiccator until further testing to minimize environmental influence.
The crosslinking characteristics of each compound were analyzed using an oscillating disc rheometer (ODR). Compounds were pre-conditioned at 23°C and 50% relative humidity for 24 hours before testing. Approximately 5g of each uncured sample was placed into the rheometer chamber and cured isothermally at 155°C. The resulting torque-time curves were used to determine the minimum torque (ML), maximum torque (MH), scorch time (t10), and optimum cure time (t90). These parameters were used to compare the crosslinking kinetics of the different formulations.
The mechanical properties of the vulcanized composites were evaluated in terms of tensile strength and abrasion resistance. Tensile tests were conducted in accordance with KS M 6518 (Rubber materials - Determination of tensile properties). Dumbbell-shaped specimens (Type 2) were prepared using a precision die cutter. Testing was performed at 23 ± 2°C using a universal testing machine at a crosshead speed of 500 mm/min. From the stress-strain data, tensile strength (kgf/cm2) and elongation at break (%) were determined. Each measurement was repeated three times, and average values were reported.
Abrasion resistance was evaluated following the KS M 6625 standard (Rubber - Determination of abrasion resistance - NBS method). A standard NBS Abrader was used. Specimens were subjected to defined loading and rotation conditions, and the resulting wear was quantified by measuring relative abrasion loss compared to a reference compound (%).
The elastic recovery behavior of the vulcanized composites was evaluated using a circular deformation test. The circular deformation test, which shows the deformation of the rubber material visually, is a method of measuring the recovery rate after aging by deforming a straight test piece into a circle, which can be simply expressed as shown in Figure 1. Rectangular specimens (2 mm thickness, 10 mm width, 100 mm length) were cut from vulcanized sheets and formed into circular loops by joining both ends with pins, thereby inducing simultaneous tensile and compressive stress across the specimen’s cross-section. Figure 2 illustrates that bending a straight specimen into a circular configuration induces tensile stress on the outer surface and compressive stress on the inner surface, thereby creating a deformation state where both tension and compression are simultaneously applied. Figure 3 shows a circular test specimen with a circular shape. To minimize the Mullins effect caused by filler-filler interactions, each specimen was pre-cycled through five deformation-relaxation cycles prior to testing.
Two distinct recovery conditions were applied. For room temperature recovery, the deformed specimens were left at 25°C for 24 hours to observe spontaneous shape restoration under ambient conditions. For thermal aging recovery, the circularly deformed specimens were placed in a forced convection oven at 80°C for 24 hours, simulating long-term heat exposure.
After the designated recovery periods, the pins were removed, and the specimens were allowed to relax freely. Figure 4 shows the result of removing the pin with both ends fixed after aging. The straight-line distance between the two ends was then measured to calculate the recovery ratio (R, %) according to Equation (1):

Where d is the recovered straight-line length, and l is the original specimen length prior to deformation. All recovery tests were conducted in triplicate under identical conditions, and average values were used for evaluation.
Results and Discussion
Analysis of curing behavior using an oscillating disc rheometer (ODR) revealed that, as depicted in Figure 5, the maximum torque (MH) in formulations containing thermoplastic resin (PVC) and plasticizer (#2~#6) decreased significantly to approximately 50~57% compared to the 100% rubber formulation (#1). This indicates a decreased proportion of rubber participating in sulfur crosslinking due to the dilution effect from non-reactive thermoplastic components.
In addition, the optimum cure time (t90) was extended by more than 10% in the thermoplastic-blended formulations. It was determined that the viscosity of the compound was reduced due to a decrease of more than 40% in crosslinking sites by sulfur. Apparent crosslink density measurements also revealed that sulfur-based crosslinks predominantly formed within the rubber phase, as the blends exhibited up to 30% higher apparent crosslink density compared to the 100% rubber formulation, despite the lack of chemical crosslinking within the PVC phase.7,8
Due to the difference in the content of accelerator B, #5 and #6 exhibit distinct crosslinking structural differences. The ODR results in Table 2 show that #5 exhibits higher maximum torque (MH) and ΔTorque than #6, indicating that it formed a denser and more stable crosslinking network within the same thermoplastic blend composition. Conversely, #6, with its excessive addition of accelerator B, exhibits a faster initial vulcanization reaction. However, during the crosslinking growth process, the proportion of long, flexible, polysulfide (C–Sn–C, n≥3) bonds significantly increases, rather than the short, strong, monosulfide (C–S–C) bonds. This is because accelerator B increases the degree of sulfur chain involvement, thereby inducing a crosslinking structure that increases the number of S–S bonds (n). Monosulfide bonds have high bonding energy and excellent thermal and oxidative stability, imparting high heat resistance and structural rigidity to the rubber network. On the other hand, polysulfide crosslinks offer high elastic resilience while tolerating large deformations under low and room temperature conditions due to their long bond lengths and high flexibility. However, during thermal aging, S–S bonds are easily cleavage and rearranged, resulting in increased irreversible deformation. Therefore, #5, with its low accelerator B content, has a relatively high proportion of monosulfides, forming a stable rubber network. On the other hand, #6, with its high accelerator B content, increases bond flexibility due to the increased polysulfidic crosslinking ratio, but has lower network stability and a structure vulnerable to thermal deformation.
As shown in Figure 6, the mechanical performance of the vulcanized composites was evaluated by measuring tensile strength and abrasion resistance. Comparing the mechanical strength of the vulcanized rubber, the blend composition of the thermoplastic-blended formulations exhibited a high performance of 20% to 50%. Notably, within these blends, a progressive decrease in mechanical strength by approximately 5% to 10% was observed as the plasticizer content increased. The #2 blend showing over 50% enhancement in abrasion resistance compared to the rubber-only control (#1). This improvement is attributed to the dispersion of PVC within the rubber matrix and the toughening effect of the thermoplastic phase.
However, an increase in plasticizer content led to a gradual decline in tensile strength, likely due to the disruption of the rubber crosslink network and the softening effect of the plasticizer. Furthermore, formulations with reduced vulcanization accelerator content (#5, #6) exhibited decreased crosslink density, resulting in lower hardness and diminished wear resistance.
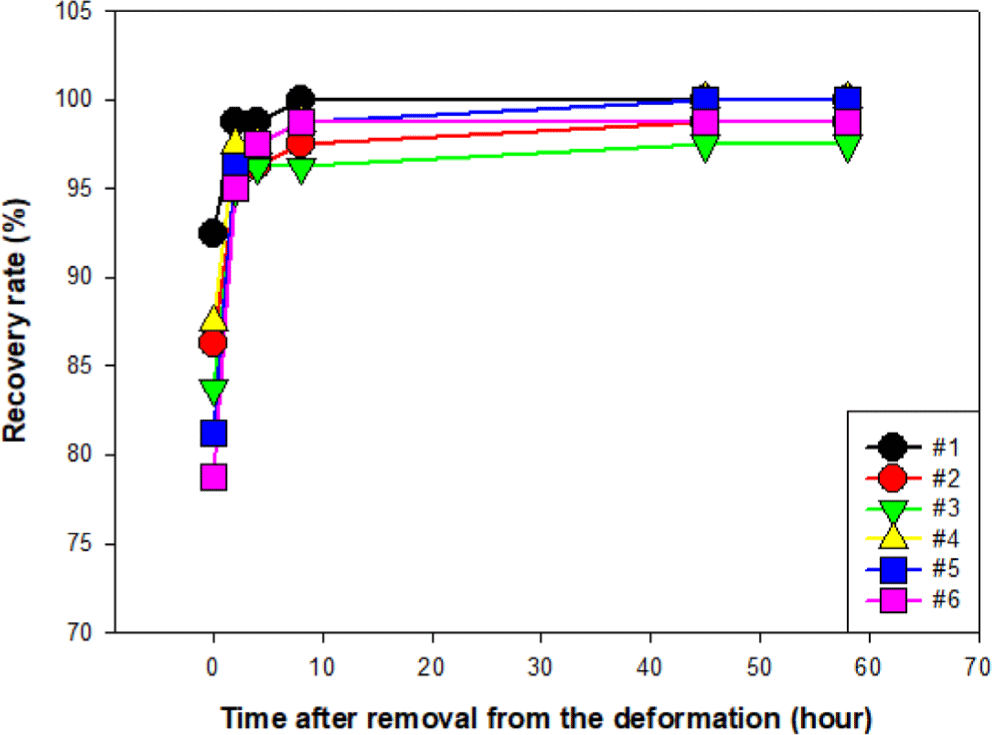
The shape recovery behavior of the specimens was examined via a circular deformation test under room temperature and thermal aging conditions. Figure 7 shows that, at room temperature (25°C), the 100% rubber formulation (#1) exhibited the highest initial recovery rate of 92.5% and reached full recovery within 8 hours.9-12 In contrast, thermoplastic-blended samples showed lower initial recovery (78.7~86.2%) and required up to 45 hours for full restoration.
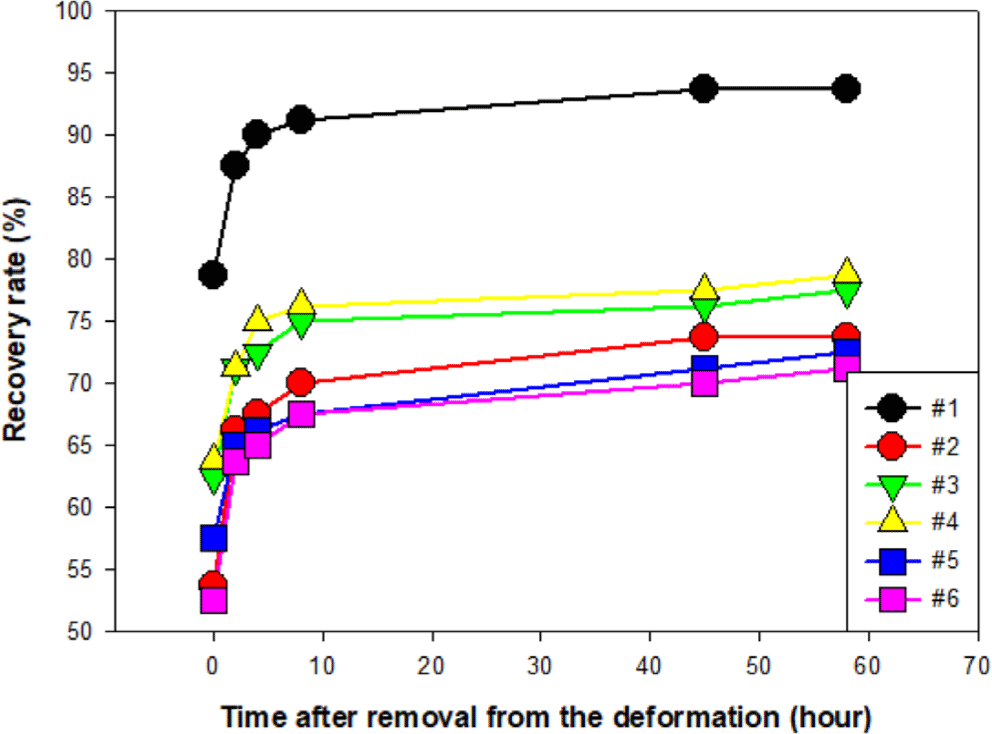
Under thermal aging conditions at 80°C for 24 hours, all formulations exhibited some degree of irreversible deformation. As shown in Figure 8, after 58 hours, the 100% rubber specimen recovered up to 93.7% but remained partially deformed thereafter. In contrast, the initial recovery of the thermoplastic-blended formulations ranged from 52.5~63.7%, and their final recovery levels after 58 hours were 71.2~78.0%. The presence of a non-crosslinked thermoplastic phase and plasticizer likely reduced recovery performance by increasing material flow and reducing elastic memory under heat.13
These findings suggest that while thermoplastic blending improves mechanical strength and abrasion resistance, it compromises thermal shape recovery. The trade-off between mechanical durability and elastic recovery under thermal stress must be carefully balanced when designing rubber-thermoplastic composites for heat-sensitive applications.14,15 Depending on the difference in the content of accelerator B, the crosslinking morphology of #5 and #6 shifts from short, strong, monosulfide-based bonds to long, flexible, polysulfide-based bonds. In #5, the lower content of accelerator B results in a relatively high monosulfide content, and the short crosslinks impart bond strength and thermal stability to the rubber chains. These structural characteristics ensure stable elastic energy storage within the rubber network even in the presence of the non-crosslinked PVC/DOP phase, effectively demonstrating chain resilience after stress relaxation in both room-temperature and high-temperature recovery tests. Therefore, #5 is structurally advantageous in both initial and long-term recovery rates.
Conversely, the increased content of accelerator B in #6 results in a higher polysulfidic crosslinking ratio, which creates a network with longer bond lengths and greater flexibility, but with lower thermal stability. This polysulfidic crosslinked structure allows relatively large elastic deformation at room temperature. However, under thermal aging conditions (80°C), S–S bonds readily cleave and rearrange, loosening the network. This, combined with increased fluidity in the thermoplastic (PVC/DOP) phase, leads to strain fixation. This results in increased irreversible deformation, and the recovery rate remains lower compared to #5. The deformation behavior of #5 and #6 is shown in Figure 9.
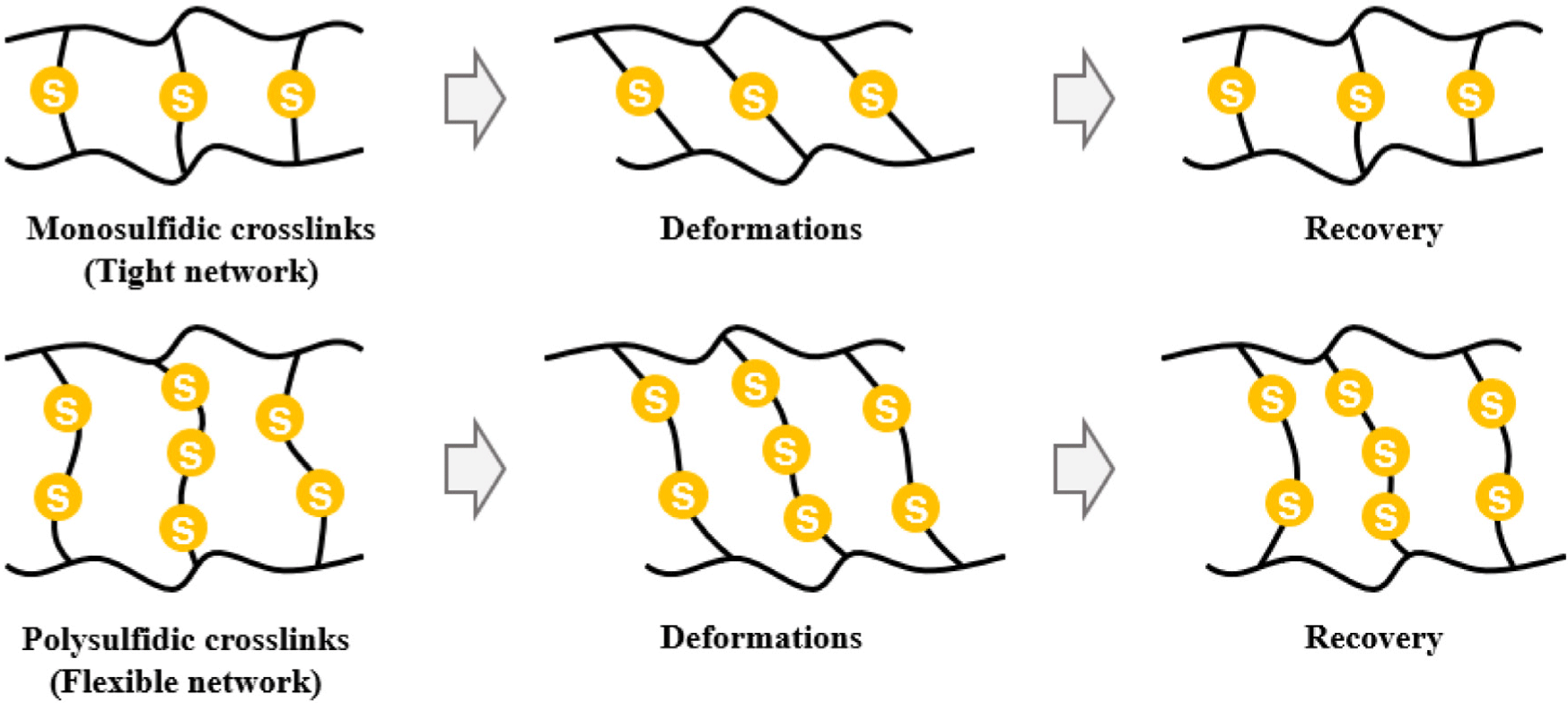
Consequently, the difference in recovery behavior between #5 and #6 is not simply due to differences in crosslink density, but rather to structural differences between the stable, heat-resistant network centered on monosulfides versus the flexible, but heat-vulnerable network centered on polysulfides. This difference is interpreted as a key mechanism determining the maintenance of shape resilience under temperature conditions.
Conclusions
This study investigated the crosslinking behavior, mechanical properties, and shape recovery characteristics of rubber/thermoplastic resin composites formulated with varying contents of plasticizer and vulcanization accelerators. The following conclusions were drawn:
The incorporation of thermoplastic resin (PVC) and plasticizer into the rubber matrix reduced the overall torque and increased the optimum cure time (t90), indicating a decrease in effective crosslinking density. Crosslinks predominantly formed within the rubber phase, while the thermoplastic phase remained physically blended without chemical crosslinking.
Monosulfidic crosslinks, with their short, strong bonds, enhance the thermal stability and resilience of the network. Polysulfidic crosslinks, on the other hand, possess long, flexible bonds, resulting in excellent initial elasticity. However, these bonds are easily broken and rearranged during thermal aging, increasing irreversible deformation. Ultimately, the difference in resilience is determined by the differences in stability and flexibility between these two crosslinking structures.
Thermoplastic-blended formulations exhibited enhanced mechanical strength and abrasion resistance, particularly in compositions with moderate plasticizer content. However, increasing the plasticizer led to a gradual reduction in mechanical strength and abrasion resistance due to the softening and dilution of the crosslinked rubber network.
At room temperature, the 100% vulcanized rubber formulation showed the fastest and most complete recovery, achieving full shape restoration within 8 hours. In contrast, thermoplastic-blended samples required up to 45 hours for full recovery. Under thermal aging at 80°C, all specimens exhibited residual deformation. After 58 hours, the rubber-only formulation recovered up to 93.7%, while thermoplastic blends showed reduced recovery (71.2~78.0%).
Monosulfidic vulcanization forms short, strong crosslinks, enhancing the thermal stability and structural rigidity of the rubber network and maintaining excellent shape resilience even after deformation. Conversely, polysulfidic vulcanization offers the advantages of long bond lengths and high flexibility. However, during thermal aging, S–S bonds are easily cleavage and rearranged, loosening the network and increasing irreversible deformation.
In summary, thermoplastic blending improves mechanical durability and abrasion resistance but compromises thermal recovery performance due to the absence of chemical crosslinking in the thermoplastic phase. These results suggest that thermoplastic resin blends enhance mechanical strength and wear resistance, making them suitable for applications subjected to repeated loading at medium and low temperatures, such as shoe outsoles, industrial cushioning materials, and durable goods for daily life and sports. However, the reduced shape recovery performance at high temperatures limits their application in applications requiring long-term high-temperature stability, such as automotive engine room components and sealants for high-temperature industrial equipment. Therefore, this suggests that rubber/thermoplastic composites should be designed to suit the end-use environment.